Chair of the Digestive Disease & Surgery Institute
Cleveland Clinic, Cleveland
This month, we look at two examples of how to manage postoperative inflammatory bowel disease, specifically recurrence of Crohn’s disease and inflammatory pouch conditions.
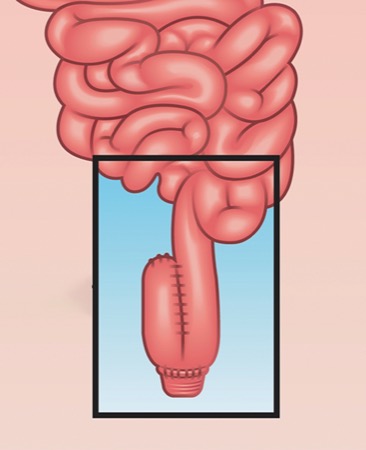
The first paper is a new clinical practice guideline from the American Gastroenterological Association about the management of pouchitis and inflammatory bowel disorders. (Note: My colleague Benjamin Cohen, MD, the clinical director for inflammatory bowel diseases at Cleveland Clinic, is one of the guideline authors.) As the authors note, almost half of the patients who undergo a restorative proctocolectomy with ileal pouch–anal anastomosis experience pouchitis within two years of the operation, and 80% develop pouchitis sometime afterward.
The new guidelines draw on literature reviews and clinical experience to give recommendations about pouchitis care, and I think they will be helpful to health providers who manage this very common postoperative complication. Along with providing detailed guidance on when to use antibiotics for the treatment of pouchitis, the guidelines also break new ground, in my view, by noting that some cases of pouchitis are more pronounced and refractory to antibiotics—in essence an “IBD of the pouch.” These scenarios will require advanced therapies beyond just antibiotics.
The other study I highlight examines predictors of Crohn’s disease recurrence after surgery and, importantly, does so prospectively rather than by relying on insurance claims data or other retrospective data sources, which have been the common type of data in used postoperative studies. It is part of a broader study that looks at genomic, microbiological and dietary drivers of Crohn’s disease.
Some of the recurrence predictors are familiar. We know already that smoking cigarettes will make someone more likely to experience Crohn’s disease again after surgery and that anti–tumor necrosis factor (TNF) medications reduce the likelihood of recurrence. Some of the predictors are new, and some refute our prior understanding, that is, penetrating disease and recurrent surgery may not present as great a risk as we once thought.
Also, the investigators show that even small amounts of inflammation after surgery, measured by the standard Rutgeerts score (i1), could be a warning of Crohn’s disease recurrence. Therefore, we should monitor individuals with any inflammatory lesions more closely than we used to, perhaps through fecal calprotectin tests, intestinal ultrasound or more frequent colonoscopies. I think the study is very well done, with compelling implications for how we care for people with Crohn’s disease undergoing surgery, and that it revises our understanding of the risk factors for postoperative recurrence.
AGA’s Take on Pouchitis
Gastroenterology 2024;166(1):59-85
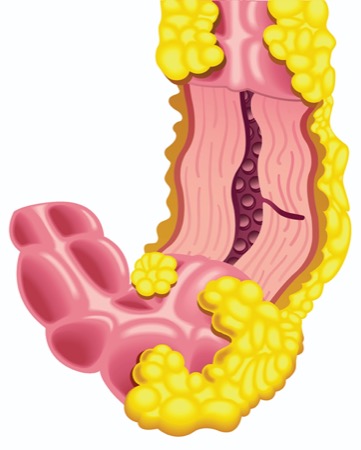
The guidelines offer different treatments for the prevention and treatment of pouchitis, Crohn’s-like disease of the pouch and cuffitis.
The authors recommend against antibiotics for primary prevention and do not recommend preventive probiotics at this time, but call for more research about whether probiotics can prevent pouchitis in patients with ulcerative colitis after proctocolectomy with ileal pouch–anal anastomosis.
For treatment of pouchitis, patients who have infrequent symptoms should receive antibiotics, the authors wrote, with ciprofloxacin and/or metronidazole as the preferred antibiotics. For patients who have recurrent pouchitis but respond to antibiotics, they recommend adding a probiotic to help reduce recurrence. If recurring patients relapse shortly after stopping antibiotics, the authors recommend long-term use of the lowest effective dose of antibiotic therapy and intermittent gap periods or use of cyclical antibiotics. These patients also may benefit from biologics or oral small molecules, they wrote.
These advanced therapies, along with corticosteroids, also are recommended for pouchitis patients who are refractory to antibiotics, as well as for patients who develop Crohn’s-like disease of the pouch. For patients who develop cuffitis, the authors suggest advanced therapies as well as topical mesalamine and topical corticosteroids.
Prospective Look at Crohn’s Recurrence
J Crohns Colitis 2023 Nov 17. doi:10.1093/ecco-jcc/jjad186
The investigators examined 365 patients with Crohn’s disease, who underwent 674 colonoscopies after surgical resection. A majority of the population identified as male (51.8%), and 10% identified as non-white. The median age was 31 years. All patients had at least one colonoscopy, 208 had two and 101 had three or more.
The researchers identified factors that led to an increased risk for endoscopic recurrence by the time of the first colonoscopy (early recurrence), or recurrence after two or more colonoscopies (late recurrence). Endoscopic recurrence was a Rutgeerts score of at least i2.
At first colonoscopy, the following attributes were associated with an increased risk for recurrence: male gender (odds ratio [OR], 1.95; 95% CI, 1.12-3.40), non-white ethnicity (OR, 2.48; 95% CI, 1.09-5.63), longer interval between surgery and colonoscopy (OR, 1.09; 95% CI, 1.002-1.18), and postoperative smoking (OR, 2.78; 95% CI, 1.16-6.67). However, prophylactic use of anti-TNF medications was associated with a reduced risk for recurrence (OR, 0.28; 95% CI, 0.14-0.55).
When patients without recurrence at their first colonoscopy were viewed at second or later colonoscopies, a Rutgeerts score of i1 at the first colonoscopy was highly associated with subsequent endoscopic recurrence (OR, 4.43; 95% CI, 1.73-11.35).
This article is from the March 2024 print issue.