The American Gastroenterological Association has released an updated guideline on pharmacologic management of adult outpatients with ulcerative colitis that, for the first time, will use a “living guideline” framework. Like the 2020 version of the guideline, the new one underscores the importance of initiating advanced therapy in place of step-up therapy with 5-aminosalicylates for treatment of moderate to severe disease but now expands the list of first-line advanced therapies recommended from six drugs to 10.
New additions to the list include the Janus kinase (JAK) inhibitor upadacitinib (Rinvoq, AbbVie), the sphingosine 1-phosphate receptor modulators ozanimod (Zeposia, Bristol Myers Squibb) and etrasimod (Velsipity, Pfizer), and the interleukin-23 inhibitors risankizumab (Skyrizi, AbbVie) and guselkumab (Tremfya, Janssen). Many of the new additions to the list were not FDA approved when the last guideline was written.
“The updated AGA Living Clinical Practice Guideline on UC provides guidance on several of the more recently approved advanced therapies and contains several valuable updates on the positioning of these therapies, both among bio-naive and bio-exposed patients with moderate to severe ulcerative colitis,” Dana J. Lukin, MD, PhD, the clinical director of the Jill Roberts Center for Inflammatory Bowel Disease at NewYork–Presbyterian Hospital/Weill Cornell Medicine, in New York City, told Gastroenterology & Endoscopy News. “This includes patients with moderate to severe ulcerative proctitis, as well as those with clinically mild disease with predictors of an adverse disease course,” added Dr. Lukin, who was not involved with the guideline.
Although the authors noted that there are still gaps in the data comparing the efficacy of different advanced therapies, lead author Siddharth Singh, MD, told Gastroenterology & Endoscopy News that it made sense to update the 2020 guideline with the new living guideline approach because so much has changed in the field in four years and continuing change is expected. “Since the field of IBD is moving rapidly with new medications and head-to-head trials that inform treatment positioning and strategy coming out at major meetings, the AGA decided to implement this approach for IBD first,” said Dr. Singh, an associate professor at the University of California, San Diego School of Medicine. “We want this to be useful for all stakeholders—patients, providers, policy developers, insurance companies, etc.—so they have the latest guidance available at all times.”
Dr. Lukin said he believed that the guideline will be useful, specifically to streamline payor acceptance of new drugs, which can be a barrier to treatment. “Overall, these updates will likely help payors make formulary decisions and can be used to justify more nuanced decision-making around initial and later-line advanced therapies,” such as “the very recently approved IL-23–selective antagonists as well as S1P receptor modulators and three JAK inhibitors.”
Changing Drug Choices
The authors of the living guideline strongly recommended 10 drugs for adult outpatients with moderate to severe UC over no treatment (Table). Three additional drugs are now under a new category of conditionally recommended over no treatment. Adalimumab dropped from a strongly recommended medication to a conditionally recommended one.
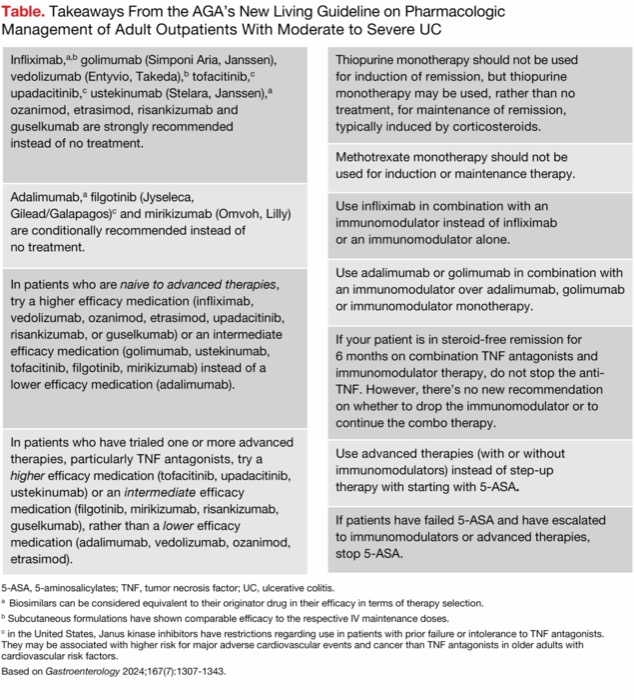
| Table. Takeaways From the AGA’s New Living Guideline on Pharmacologic Management of Adult Outpatients With Moderate to Severe UC |
| Infliximab,a,b golimumab (Simponi Aria, Janssen), vedolizumab (Entyvio, Takeda),b tofacitinib,c upadacitinib,c ustekinumab (Stelara, Janssen),a ozanimod, etrasimod, risankizumab and guselkumab are strongly recommended instead of no treatment. |
| Adalimumab,a filgotinib (Jyseleca, Gilead/Galapagos)c and mirikizumab (Omvoh, Lilly) are conditionally recommended instead of no treatment. |
| In patients who are naive to advanced therapies, try a higher efficacy medication (infliximab, vedolizumab, ozanimod, etrasimod, upadacitinib, risankizumab, or guselkumab) or an intermediate efficacy medication (golimumab, ustekinumab, tofacitinib, filgotinib, mirikizumab) instead of a lower efficacy medication (adalimumab). |
| In patients who have trialed one or more advanced therapies, particularly TNF antagonists, try a higher efficacy medication (tofacitinib, upadacitinib, ustekinumab) or an intermediate efficacy medication (filgotinib, mirikizumab, risankizumab, guselkumab), rather than a lower efficacy medication (adalimumab, vedolizumab, ozanimod, etrasimod). |
| Thiopurine monotherapy should not be used for induction of remission, but thiopurine monotherapy may be used, rather than no treatment, for maintenance of remission, typically induced by corticosteroids. |
| Methotrexate monotherapy should not be used for induction or maintenance therapy. |
| Use infliximab in combination with an immunomodulator instead of infliximab or an immunomodulator alone. |
| Use adalimumab or golimumab in combination with an immunomodulator over adalimumab, golimumab or immunomodulator monotherapy. |
| If your patient is in steroid-free remission for 6 months on combination TNF antagonists and immunomodulator therapy, do not stop the anti-TNF. However, there’s no new recommendation on whether to drop the immunomodulator or to continue the combo therapy. |
| Use advanced therapies (with or without immunomodulators) instead of step-up therapy with starting with 5-ASA. |
| If patients have failed 5-ASA and have escalated to immunomodulators or advanced therapies, stop 5-ASA. |
| 5-ASA, 5-aminosalicylates; TNF, tumor necrosis factor; UC, ulcerative colitis. a Biosimilars can be considered equivalent to their originator drug in their efficacy in terms of therapy selection. b Subcutaneous formulations have shown comparable efficacy to the respective IV maintenance doses. c in the United States, Janus kinase inhibitors have restrictions regarding use in patients with prior failure or intolerance to TNF antagonists. They may be associated with higher risk for major adverse cardiovascular events and cancer than TNF antagonists in older adults with cardiovascular risk factors. Based on Gastroenterology 2024;167(7):1307-1343. |
Of the 10 strongly recommended agents, upadacitinib has been shown to have a larger desirable effect on clinical remission, the authors noted. In analyzing the literature, they found upadacitinib had a clinical meaningful difference from placebo of more than 20%—above the 10% threshold of the other nine drugs listed. The three drugs conditionally recommended did not meet that 10% threshold.
The guideline authors used a network meta-analysis approach to compare the available drug literature for patients who are bio-naive and for those who are bio-exposed. In the second recommendation, they determined that seven drugs could be considered to have higher efficacy and should be strongly recommended for patients who have not taken advanced therapies: infliximab, vedolizumab, ozanimod, etrasimod, upadacitinib, risankizumab and guselkumab. For patients who have been exposed to advanced therapies, as discussed in the third recommendation, the list of strongly recommended drugs shortens to three: tofacitinib, upadacitinib and ustekinumab (Stelara, Janssen).
“We have utilized best available evidence to inform positioning of different advanced therapies in biologic-naive and biologic-exposed patients with ulcerative colitis,” Dr. Singh said. “To do this, we have bucketed medications in terms of relative efficacy. At the same time, we wanted this to be simple to disseminate and implement. Recognizing nuances in day-to-day practice and specific situations, we wanted patients and providers to make the best decision for the patients’ health. Hence, while making these buckets, we were deliberate in ensuring we represent medications of different classes, routes of administration, safety considerations, speed of onset, etc., within each bucket.”
The recommendations also leave room for personalization, with nods to various drug classes that may be better for some patients over others. For instance, the authors noted that the anti-integrin vedolizumab and the anti-interleukin drugs may be better options for certain patients because they may be associated with lower rates of infections than anti–tumor necrosis factor (TNF) drugs. They also noted that data vary related to drug use in pregnancy.
The guideline authors also noted that extended induction regimens (up to 16 weeks) or dose escalation may be beneficial for some patients, especially those with severe disease.
Dr. Lukin said this provides “helpful nuance” and noted that the authors mentioned that clinicians should take into account possible safety concerns with higher doses. In addition, “they provide guidance that some medications, such as ozanimod, which are effective for bio-naive and singly exposed patients, may require a longer duration for induction of remission among multiply biologic-exposed patients,” Dr. Lukin explained.
Knowledge Gaps
Although the field has exploded with different drug classes and new data, the authors were quick to point out that knowledge gaps remain in many areas. Besides the aforementioned gaps in head-to-head trials, there also are data needed on drug choices after anti-TNF failure and what to do when anti-TNF and immunomodulator combination therapy leads to remission—can the immunomodulator be dropped or should it be continued?
Furthermore, this guideline focuses on standards of clinical remission but does not touch on endoscopic remission or other trial outcomes. The authors noted that the current literature is too mixed on these outcomes to make uniform decisions. “As end points begin to incorporate endoscopic and histologic healing, consistency in reporting outcomes across trials will be important to inform relative positioning,” they wrote. “However, it will remain a challenge to compare older trials that may not have assessed this outcome or differed in their study design.” With these changes in the field, the authors also anticipated that biomarkers will begin to play more of a role in choosing which drug is right for which patient.
But that flexibility to change will be a new component now that it has transitioned to a “living guideline.” The AGA said it will be updated every six months, which makes sense for certain areas where drug development is occurring rapidly, Dr. Singh said. “For other topics, we will explore variations of this ‘living’ approach to make all guidelines timely.”
The goal is to help gastroenterologists help their patients, Dr. Singh said. “We believe guidelines should be living documents to allow best available evidence to be disseminated in a timely manner to facilitate best care for patients.”
—Meaghan Lee Callaghan
Dr. Singh reported a financial relationship with Pfizer. Dr. Lukin reported financial relationships with AbbVie, Boehringer Ingelheim, Bristol Myers Squibb, Fresenius Kabi, Janssen, Lilly, Pfizer and Takeda. He is a member of the Gastroenterology & Endoscopy News editorial board.
This article is from the January 2025 print issue.