Digestive Health Clinical Research Center
Director of Neurogastroenterology/Motility
Director, Digestive Health Clinical Research Center
Augusta, Georgia
Constipation is a common gastrointestinal problem afflicting 15% of people of all ages worldwide.1 Characterized by difficult, less frequent, or incomplete passage of bowel movements, it leads to symptoms including straining, hard stools, use of digital maneuvers, incomplete evacuation, bloating, and abdominal pain. Constipation poses a significant healthcare burden. The 2020 estimate of over-the-counter laxatives alone was approximately $1.5 billion.2 About one-third of constipated patients also suffer from anxiety and depression.3
The condition is broadly classified as primary constipation—pathophysiologic dysfunction of the colon and rectum due to the overlapping subtypes slow-transit constipation, dyssynergic defecation, and irritable bowel syndrome with constipation (
Although over-the-counter treatments such as fiber supplements, laxatives, and dietary changes have been the mainstay for managing mild constipation, patients with chronic symptoms may respond better to prescription second-tier drugs such as linaclotide (Linzess, AbbVie), lubiprostone, plecanatide (Trulance, Salix), and prucalopride (Motegrity, Takeda).2 However, many patients are unable to take these drugs because of adverse effects, costs, or insurance constraints. Several emerging or recently approved treatments can offer more effective and targeted relief of symptoms. In this review article, we explore the latest developments related to nondrug treatments for constipation.
Kiwifruit
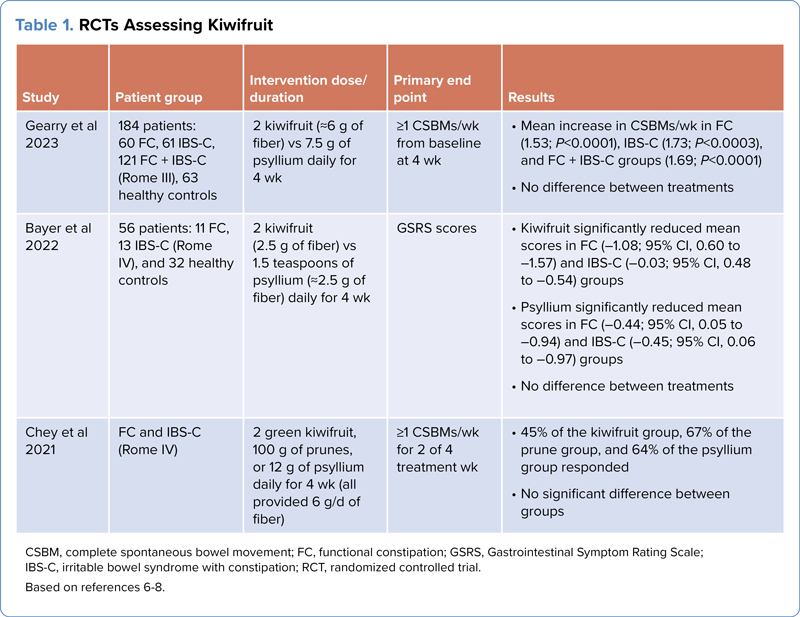
Although the importance of a high-fiber diet/supplement to control constipation-related symptoms has been well established, recent randomized controlled trials (RCTs) on kiwifruit have shown that eating 2 kiwifruit per day may be beneficial for patients with constipation (Table 1).6-8 In a recent study, Gearry et al compared kiwifruit with psyllium in 184 patients with functional constipation or
| Table 1. RCTs Assessing Kiwifruit | ||||
| Study | Patient group | Intervention dose/duration | Primary end point | Results |
|---|---|---|---|---|
| Gearry et al 2023 | 184 patients: 60 FC, 61 | 2 kiwifruit (˜6 g of fiber) vs 7.5 g of psyllium daily for 4 wk | =1 CSBMs/wk from baseline at 4 wk |
|
| Bayer et al 2022 | 56 patients: 11 FC, 13 | 2 kiwifruit (2.5 g of fiber) vs 1.5 teaspoons of psyllium (˜2.5 g of fiber) daily for 4 wk | GSRS scores |
|
| Chey et al 2021 | FC and | 2 green kiwifruit, 100 g of prunes, or 12 g of psyllium daily for 4 wk (all provided 6 g/d of fiber) | =1 CSBMs/wk for 2 of 4 treatment wk |
|
| CSBM, complete spontaneous bowel movement; FC, functional constipation; GSRS, Gastrointestinal Symptom Rating Scale; Based on references 6-8. | ||||
Vibrating Capsules
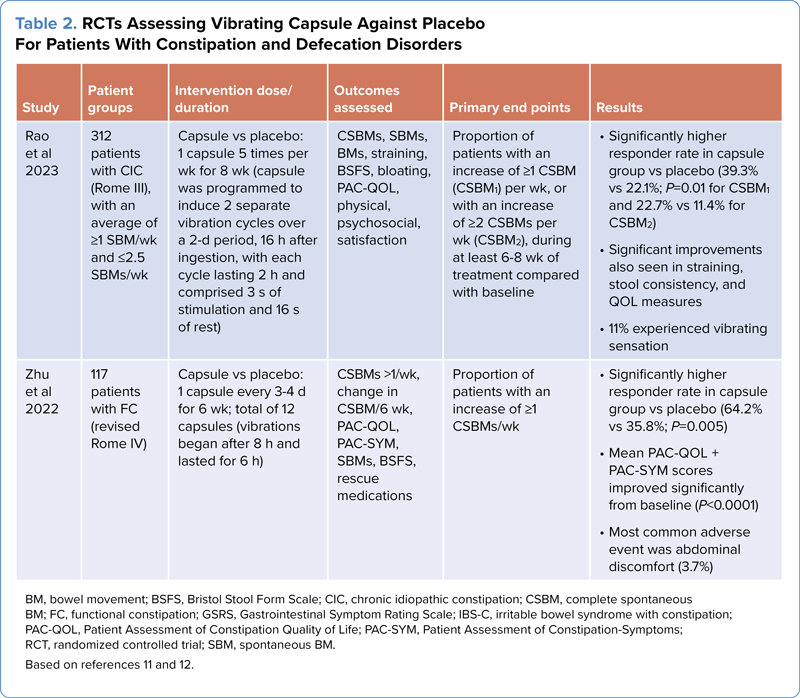
An innovative new approach to address constipation has been the development of a vibrating capsule (Vibrant) that helps to mechanically stimulate the intestines directly and induce local peristalsis and augment the natural circadian rhythm.9,10 The capsule can be programmed to vibrate according to a fixed schedule, and the number of vibrations also can be adjusted. Patients reported that CSBMs occur closely with the scheduled vibrations of the pill (Table 2).9,10
| Table 2. RCTs Assessing Vibrating Capsule Against Placebo For Patients With Constipation and Defecation Disorders | |||||
| Study | Patient groups | Intervention dose/duration | Outcomes assessed | Primary end points | Results |
|---|---|---|---|---|---|
| Rao et al 2023 | 312 patients with CIC (Rome III), with an average of =1 SBM/wk and =2.5 SBMs/wk | Capsule vs placebo: 1 capsule 5 times per wk for 8 wk (capsule was programmed to induce 2 separate vibration cycles over a 2-d period, 16 h after ingestion, with each cycle lasting 2 h and comprised 3 s of stimulation and 16 s of rest) | CSBMs, SBMs, BMs, straining, BSFS, bloating, PAC-QOL, physical, psychosocial, satisfaction | Proportion of patients with an increase of =1 CSBM (CSBM1) per wk, or with an increase of =2 CSBMs per wk (CSBM2), during at least 6-8 wk of treatment compared with baseline |
|
| Zhu et al 2022 | 117 patients with FC (revised Rome IV) | Capsule vs placebo: 1 capsule every 3-4 d for 6 wk; total of 12 capsules (vibrations began after 8 h and lasted for 6 h) | CSBMs >1/wk, change in CSBM/6 wk, PAC-QOL, PAC-SYM, SBMs, BSFS, rescue medications | Proportion of patients with an increase of =1 CSBMs/wk |
|
| BM, bowel movement; BSFS, Bristol Stool Form Scale; CIC, chronic idiopathic constipation; CSBM, complete spontaneous BM; FC, functional constipation; GSRS, Gastrointestinal Symptom Rating Scale; Based on references 11 and 12. | |||||
In a phase 3 RCT comparing the Vibrant capsule administered 5 times per week with a sham capsule, there was significant improvement in constipation symptoms and QOL measures.9 The primary outcome measure of an increase in 1 or more CSBMs per week or at least 2 CSBMs per week was significantly higher with the Vibrant capsule than with the sham capsule. Stool consistency, straining effort, and the number of patients with at least 3 CSBMs per week also improved significantly in the Vibrant capsule group. Adverse events included a sensation of vibration, which 11% of patients reported, but none of the patients withdrew from the trial due to the sensation. No diarrhea was seen.
Another recent study showed that vibrating capsule stimulation in the gut (normal and enhanced) is perceived in the parieto-occipital region, and the gastric evoked potentials showed intensity-dependent correlation with gut perception.10 These findings suggest that the Vibrant capsule–induced evoked potentials may provide insights into how the brain processes gut sensations.10
The Vibrant capsule is approved by the FDA for treatment of chronic idiopathic constipation and could prove to be a significant advance in our approach to managing constipation. In the future, it may be possible to change the vibration settings and personalize delivery of vibration for each patient.11
Zhu et al evaluated another vibrating capsule (Vibrabot, Ankon Medical) in a phase 2 study and found that it improved the number of BMs and was safe.12 In this trial, the investigators were allowed to use 2 different vibration modes based on the patient’s tolerance.
Neuromodulation Therapies
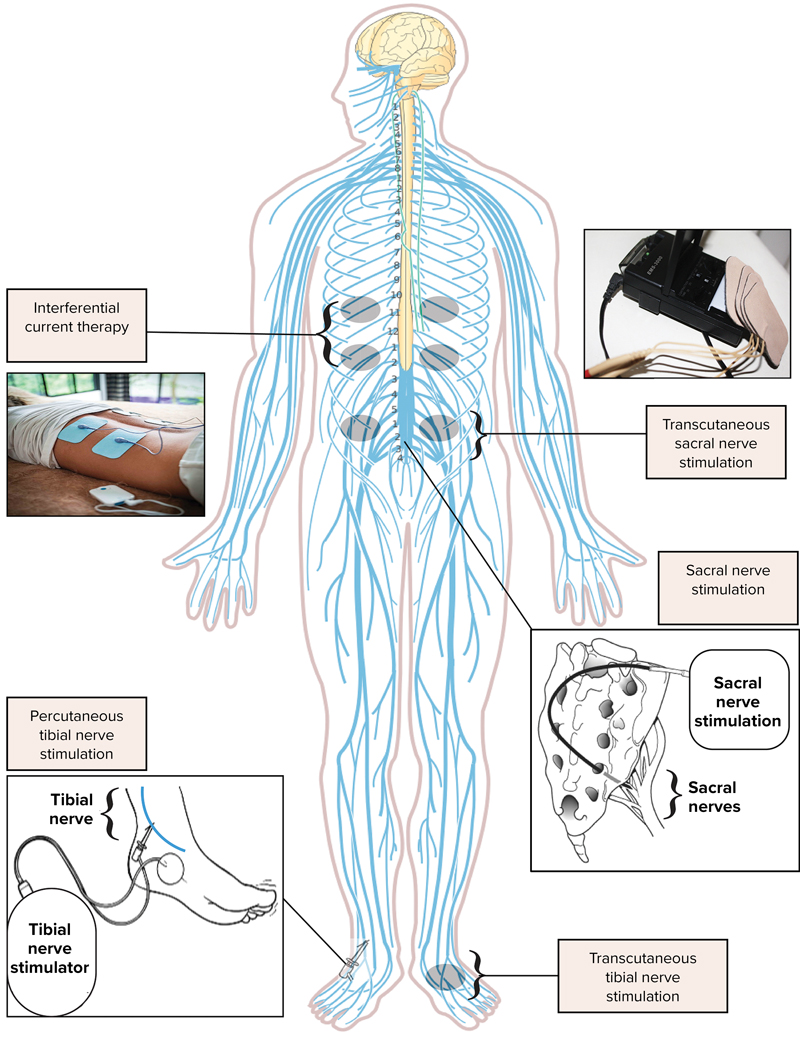
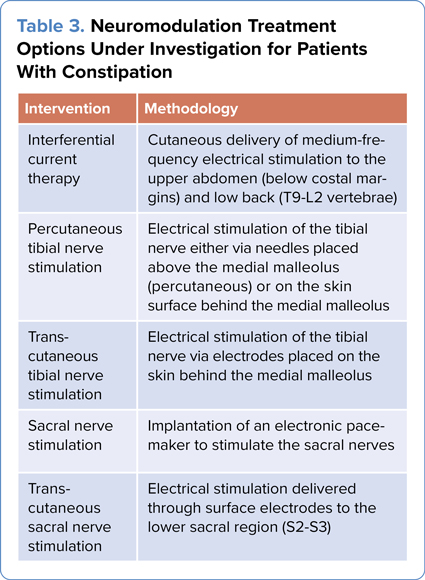
Neuromodulation is a broad term that refers to the use of various techniques to modulate or alter the activity of the nervous system. In the context of constipation, neuromodulation techniques can be employed to influence the nerve signals that control bowel function and motility. Multiple approaches to neuromodulation are under investigation, each with variable efficacy and adverse effects (Figure and Table 3).
| Table 3. Neuromodulation Treatment Options Under Investigation for Patients With Constipation | |
| Intervention | Methodology |
|---|---|
| Interferential current therapy | Cutaneous delivery of medium-frequency electrical stimulation to the upper abdomen (below costal margins) and low back (T9-L2 vertebrae) |
| Percutaneous tibial nerve stimulation | Electrical stimulation of the tibial nerve either via needles placed above the medial malleolus (percutaneous) or on the skin surface behind the medial malleolus |
| Trans-cutaneous tibial nerve stimulation | Electrical stimulation of the tibial nerve via electrodes placed on the skin behind the medial malleolus |
| Sacral nerve stimulation | Implantation of an electronic pacemaker to stimulate the sacral nerves |
| Trans-cutaneous sacral nerve stimulation | Electrical stimulation delivered through surface electrodes to the lower sacral region (S2-S3) |
Interferential Current Therapy
Interferential current therapy (IFC) is a noninvasive and nonpharmacologic treatment that delivers medium-frequency electrical current to stimulate deep tissue nerves without activating cutaneous nerves. IFC works by producing 2 out-of-phase medium-frequency alternating currents. Since they are out of phase, the stimulations interfere with each other, creating a sinusoidal modulation, or an alternating current.13
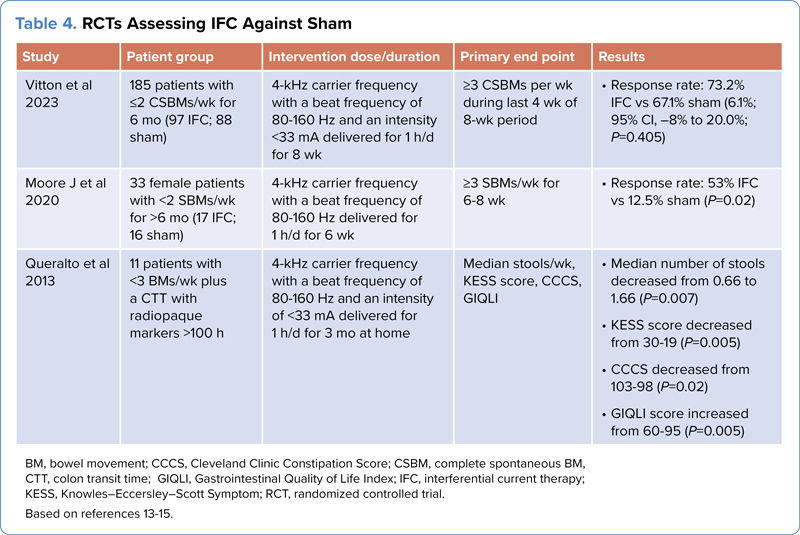
Initially studied in children, IFC has been evaluated for adult chronic constipation in 3 studies (Table 4).13-15 Vitton et al performed a phase 3 sham versus placebo trial in 185 patients and found no significant improvement in their primary end point of at least 3 CSBMs per week at the end of 8 weeks (IFC, 67.1% vs sham, 73.2%).13 They found significant improvement in the Patient Assessment of Constipation-Symptoms (PAC-SYM) questionnaire but no change in Patient Assessment of QOL (PAC-QOL). In another study comparing sham versus IFC in 33 patients, the investigators found a significant improvement in 53% (9/17) of the IFC group.14 An earlier pilot study evaluating 11 patients showed significant improvement in stool frequency per week and scores on the Knowles–Eccersley–Scott Symptom and Gastrointestinal QOL Index questionnaires.15
| Table 4. RCTs Assessing IFC Against Sham | ||||
| Study | Patient group | Intervention dose/duration | Primary end point | Results |
|---|---|---|---|---|
| Vitton et al 2023 | 185 patients with =2 CSBMs/wk for 6 mo (97 IFC; 88 sham) | 4-kHz carrier frequency with a beat frequency of 80-160 Hz and an intensity <33 mA delivered for 1 h/d for 8 wk | =3 CSBMs per wk during last 4 wk of 8-wk period |
|
| Moore J et al 2020 | 33 female patients with <2 SBMs/wk for >6 mo (17 IFC; 16 sham) | 4-kHz carrier frequency with a beat frequency of 80-160 Hz delivered for 1 h/d for 6 wk | =3 SBMs/wk for 6-8 wk |
|
| Queralto et al 2013 | 11 patients with <3 BMs/wk plus a CTT with radiopaque markers >100 h | 4-kHz carrier frequency with a beat frequency of 80-160 Hz and an intensity of <33 mA delivered for 1 h/d for 3 mo at home | Median stools/wk, KESS score, CCCS, GIQLI |
|
| BM, bowel movement; CCCS, Cleveland Clinic Constipation Score; CSBM, complete spontaneous BM; CTT, colon transit time; GIQLI, Gastrointestinal Quality of Life Index; IFC, interferential current therapy; KESS, Knowles–Eccersley–Scott Symptom; RCT, randomized controlled trial. Based on references 13-15. | ||||
Tibial Nerve Stimulation
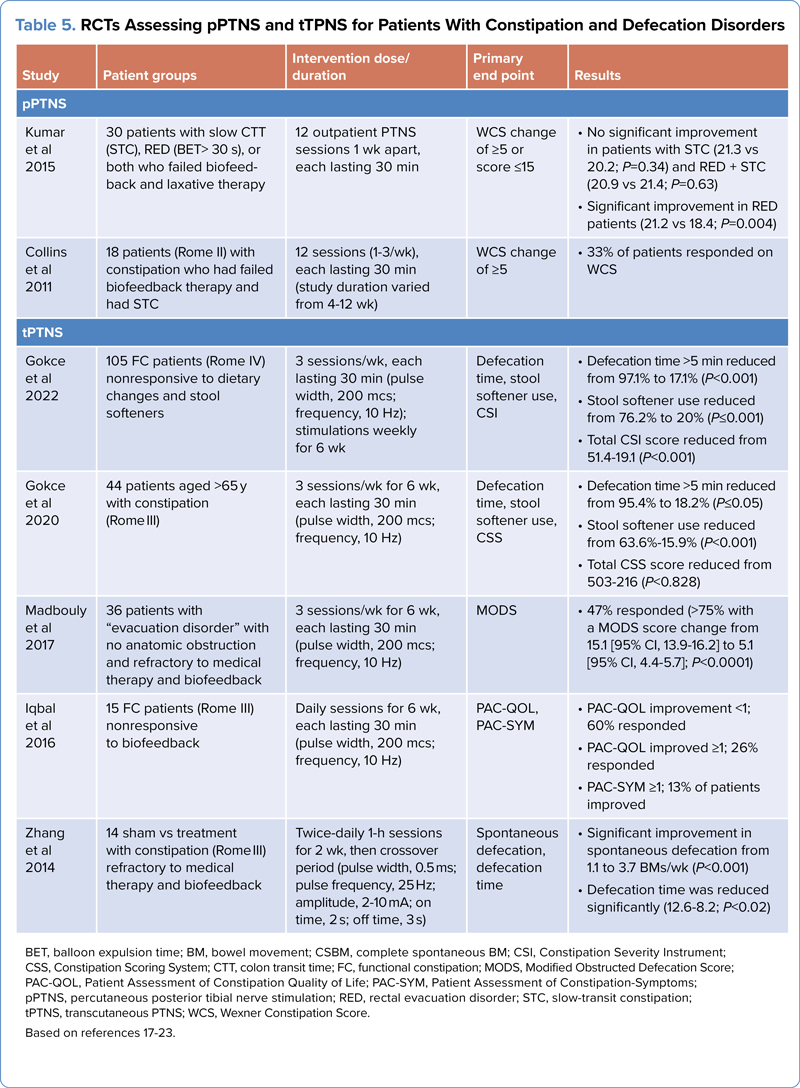
Posterior tibial nerve stimulation (PTNS) uses electrical stimulation to activate the sacral nerves retrogradely and improve bowel function.16 Percutaneous PTNS (pPTNS) involves percutaneous electrical impulses delivered to the tibial nerve using needles placed under the skin near the ankle, whereas transcutaneous PTNS (tPTNS) involves placing surface electrodes near the medial malleolus region.17 Table 5 reviews the studies on pPTNS and
| Table 5. RCTs Assessing pPTNS and tTPNS for Patients With Constipation and Defecation Disorders | ||||
| Study | Patient groups | Intervention dose/duration | Primary end point | Results |
|---|---|---|---|---|
| pPTNS | ||||
| Kumar et al 2015 | 30 patients with slow CTT (STC), RED (BET> 30 s), or both who failed biofeedback and laxative therapy | 12 outpatient PTNS sessions 1 wk apart, each lasting 30 min | WCS change of =5 or score =15 |
|
| Collins et al 2011 | 18 patients (Rome II) with constipation who had failed biofeedback therapy and had STC | 12 sessions (1-3/wk), each lasting 30 min (study duration varied from 4-12 wk) | WCS change of =5 |
|
| tPTNS | ||||
| Gokce et al 2022 | 105 FC patients (Rome IV) nonresponsive to dietary changes and stool softeners | 3 sessions/wk, each lasting 30 min (pulse width, 200 mcs; frequency, 10 Hz); stimulations weekly for 6 wk | Defecation time, stool softener use, CSI |
|
| Gokce et al 2020 | 44 patients aged >65 y with constipation (Rome III) | 3 sessions/wk for 6 wk, each lasting 30 min (pulse width, 200 mcs; frequency, 10 Hz) | Defecation time, stool softener use, CSS |
|
| -Madbouly et al 2017 | 36 patients with “-evacuation disorder” with no anatomic obstruction and refractory to medical therapy and biofeedback | 3 sessions/wk for 6 wk, each lasting 30 min (pulse width, 200 mcs; frequency, 10 Hz) | MODS |
|
| Iqbal et al 2016 | 15 FC patients (Rome III) nonresponsive to biofeedback | Daily sessions for 6 wk, each lasting 30 min (pulse width, 200 mcs; frequency, 10 Hz) | PAC-QOL, PAC-SYM |
|
| Zhang et al 2014 | 14 sham vs treatment with constipation (Rome III) refractory to medical therapy and biofeedback | Twice-daily 1-h sessions for 2 wk, then crossover period (pulse width, 0.5 ms; pulse frequency, 25 Hz; amplitude, 2-10 mA; on time, 2 s; off time, 3 s) | Spontaneous defecation, defecation time |
|
| BET, balloon expulsion time; BM, bowel movement; CSBM, complete spontaneous BM; CSI, Constipation Severity Instrument; CSS, Constipation Scoring System; CTT, colon transit time; FC, functional constipation; MODS, Modified Obstructed Defecation Score; PAC-QOL, Patient Assessment of Constipation Quality of Life; PAC-SYM, Patient Assessment of Constipation-Symptoms; pPTNS, percutaneous posterior tibial nerve stimulation; RED, rectal evacuation disorder; STC, slow-transit constipation; tPTNS, transcutaneous PTNS; WCS, Wexner Constipation Score. Based on references 17-23. | ||||
The data on pPTNS have been mixed.18,19 In 2011, Collins et al studied a cohort group of 18 patients, and found a significant improvement in the Wexner Constipation Score (WCS), colonic transit time (CTT), the PAC-QOL, and median change in BMs per week.18 However, in 2015, Kumar et al evaluated 30 patients and found no significant changes in the WCS and CTT and balloon expulsion time.19
tPTNS overcomes the invasiveness of the pPTNS method. Gokce et al evaluated the effect of tPTNS on defecation time (group 1, <5 minutes; group 2, 5-11 minutes; group 3, >11 minutes). tPTNS significantly reduced the defecation time to less than 5 minutes in the majority of patients (82.9% vs 61.9% at baseline).20 Stool softener use, Constipation Severity Instrument (CSI) scores, and anal pain also were significantly reduced. Gokce et al also previously evaluated tPTNS in a geriatric cohort with significant reduction in defecation time, CSI scores, and use of stool softeners.21 However, standard outcome measures such as the CSBM rate were not reported.
In another study of bilateral tPTNS, 47% were responders whereas 53% were nonresponders, with tPTNS resulting in no significant change in patents with functional outlet obstruction and constipation who failed medical management and biofeedback therapy.22 Iqbal et al evaluated tPTNS use in 15 patients and found no improvement in bowel symptoms but significant improvement on the PAC-QOL (<1 difference from baseline).17 However, 66% of the patients required rescue laxatives. Zhang et al conducted a sham versus tPTNS crossover trial of 12 patients and showed a significant improvement on spontaneous defecation, defecation time, and PAC-SYM parameters.23
Although these small uncontrolled studies demonstrate mixed results, larger RCTs with standard objective measures are necessary to establish the efficacy and safety of PTNS. One large multicenter RCT failed to demonstrate benefit of PTNS in fecal incontinence.24
Sacral Nerve Stimulation
Sacral nerve stimulation (SNS), or sacral neuromodulation, has been tested for chronic constipation that has not responded to conservative treatments.25 SNS involves electrical stimulation of the S2 and S3 nerves and implantation of a small device under the skin in the low back or buttock area. SNS implantation typically is done in 2 stages. In the first stage, a temporary wire is inserted to assess the patient’s response to therapy; if results are positive, a permanent device is implanted in the second stage.
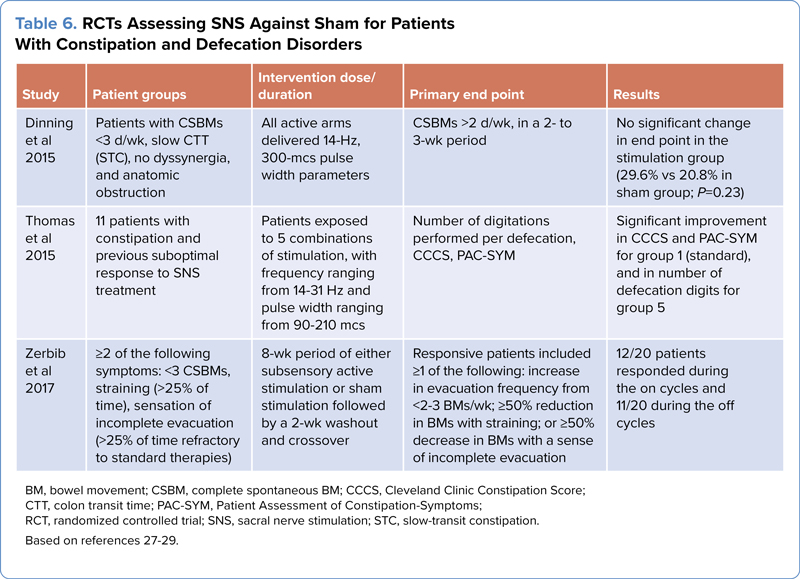
SNS has shown conflicting results (Table 6). Although a large open-label study showed significant improvements in patients with constipation refractory to other therapies,26 recent RCTs have revealed no significant improvement.27-29 In a cohort of 11 patients, Thomas et al reported improvement in 2 of 10 outcomes for patients, using different combinations of SNS stimulation for each patient and comparing with standard therapy in a crossover trial.27 Likewise, in larger RCTs, Dinning et al28 and Zerbib et al29 found no significant improvements in QOL measures, CTT, and bowel symptoms. There was a higher incidence of adverse events in these trials, namely pain (2.7%-44%), explantation of the device (8.3%), and wound infection (2.7%-20.3%).28,29 Given the costs and lack of benefit, SNS is unlikely to be approved for constipation.
| Table 6. RCTs Assessing SNS Against Sham for Patients With Constipation and Defecation Disorders | ||||
| Study | Patient groups | Intervention dose/duration | Primary end point | Results |
|---|---|---|---|---|
| Dinning et al 2015 | Patients with CSBMs <3 d/wk, slow CTT (STC), no dyssynergia, and anatomic obstruction | All active arms delivered 14-Hz, 300-mcs pulse width parameters | CSBMs >2 d/wk, in a 2- to 3-wk period | No significant change in end point in the stimulation group (29.6% vs 20.8% in sham group; P=0.23) |
| Thomas et al 2015 | 11 patients with constipation and previous suboptimal response to SNS treatment | Patients exposed to 5 combinations of stimulation, with frequency ranging from 14-31 Hz and pulse width ranging from 90-210 mcs | Number of digitations performed per defecation, CCCS, PAC-SYM | Significant improvement in CCCS and PAC-SYM for group 1 (standard), and in number of defecation digits for group 5 |
| Zerbib et al 2017 | =2 of the following symptoms: <3 CSBMs, straining (>25% of time), sensation of incomplete evacuation (>25% of time refractory to standard therapies) | 8-wk period of either subsensory active stimulation or sham stimulation followed by a 2-wk washout and crossover | Responsive patients included =1 of the following: increase in evacuation frequency from <2-3 BMs/wk; =50% reduction in BMs with straining; or =50% decrease in BMs with a sense of incomplete evacuation | 12/20 patients responded during the on cycles and 11/20 during the off cycles |
| BM, bowel movement; CSBM, complete spontaneous BM; CCCS, Cleveland Clinic Constipation Score; CTT, colon transit time; PAC-SYM, Patient Assessment of Constipation-Symptoms; RCT, randomized controlled trial; SNS, sacral nerve stimulation; STC, slow-transit constipation. Based on references 27-29. | ||||
Transcutaneous SNS
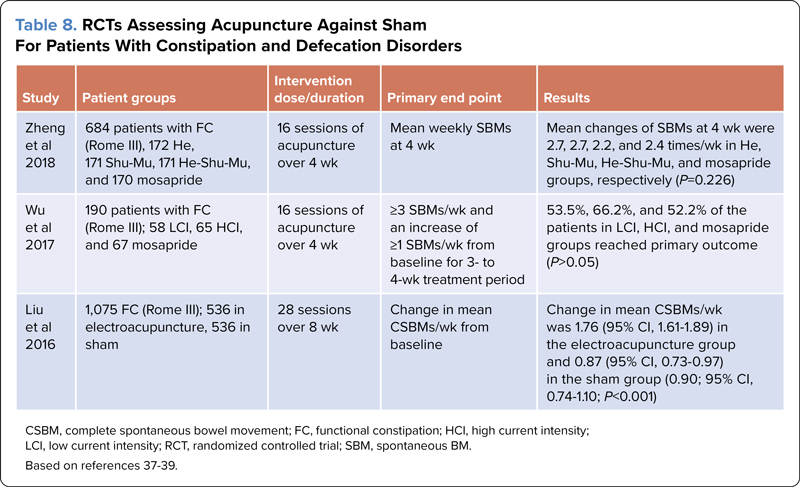
The sacral nerve also may be stimulated using surface electrodes placed near the sacral region of the spinal cord (S2-S3). RCTs with a small sample size (<30 patients) have been conducted with mixed results (Table 7).30-32 With a 6-week treatment protocol and cohort of 15 participants, Kim et al found transcutaneous SNS (tSNS) to significantly increase BMs per week and the Constipation Assessment Scale (CAS) score.30 However, the inclusion criteria for participants were not based on standard diagnosis. Iqbal et al evaluated a cohort of 16 patients and found no significant changes in PAC-SYM parameters, stool frequency, Cleveland Clinic Constipation Score, and the PAC-QOL.31 However, the treatment protocol differed from that used by Kim et al. Lastly, Yang et al evaluated 28 patients (14 sham and 14 tSNS) and showed a significant improvement in CAS score and number of bowel movements (BMs) per week, as well as reduced abdominal pain.32 Further studies with a larger sample sizes are required to evaluate whether tSNS can improve symptoms in chronic constipation.
| Table 7. RCTs Assessing Transcutaneous SNS Against Sham For Patients With Constipation and Defecation Disorders | ||||
| Study | Patient groups | Intervention dose/duration | Primary end point | Results |
|---|---|---|---|---|
| Yang et al 2017 | 28 women with STC | 14 patients randomized to treatment and 14 to sham; stimulation performed for 20 min 3 times/wk for 4 wk | CAS score, abdominal pain, number of BMs/wk | Significant improvement in CAS score, abdominal pain, and evacuation frequency per week |
| Iqbal et al 2016 | 16 FC patients (Rome III) | Continuous electrical stimulation (device worn continuously for 12 h/d) (frequency of 40 Hz, a pulse width of 220 mcs, work cycles of 99 s with 2 s rest) | PAC-SYM | No significant changes in PAC-SYM when documenting |
| Kim et al 2014 | 15 patients with CAS score >4 points and <3 BMs/wk | Stimulation performed for 20 min 3 times/wk for 6 wk (frequency of 50 Hz with burst intervals of 3 and 6 s) | BMs/d, BMs/wk, CAS score | Significant improvement in BMs/d and BMs/wk |
| BM, bowel movement; CAS, Constipation Assessment Scale; FC, functional constipation; PAC-SYM, Patient Assessment of Constipation Symptoms; RCT, randomized controlled trial; SNS sacral nerve stimulation; STC, slow-transit constipation. Based on references 30-32. | ||||
Exoperistalsis Treatment
Abdominal massage, specifically colon massage, may alleviate symptoms of chronic constipation. An open-label study evaluated an exoperistaltic device that delivered intermittent massage therapy for 20 minutes up to 3 times daily for 4 weeks in a cohort of 92 patients with neurogenic bowel dysfunction or idiopathic constipation.33 Patients experienced a significant improvement in the number of complete BMs per week, with reduced use of laxatives and improved QOL measures. However, CTT and stool consistency were unchanged. Although exoperistalsis may seem like a promising treatment for constipated patients, further RCT studies are required to establish efficacy, particularly compared with a sham device.
Acupuncture is an ancient Chinese medical practice that involves inserting thin needles into specific points on the body. Multiple studies suggest that traditional or electroacupuncture may help improve bowel function and alleviate constipation symptoms.34 However, these studies evaluate different sites and regimens and modes of treatment (ie, electrical vs traditional), producing considerable heterogeneity within the studies.
Jie et al and Zhang et al reviewed studies assessing electroacupuncture.35,36 Jie et al’s review included studies that had substantial heterogeneity and evaluated different etiologies of constipation, such as post-stroke constipation, diabetic constipation, opioid-induced constipation, and constipation in patients with Parkinson’s disease.35 Of the 13 studies in Jie et al’s review, 2 were analyzed and showed a significant improvement in CSBMs, with a mean difference of 0.42 (P=0.001) and I2, 0 (P=0.58). Zhang et al’s meta-analysis included 5 RCTs that only evaluated use of electroacupuncture, with similar sites of puncture, in patients with functional constipation.36 They showed a significant improvement in CSBMs per week at 4 weeks, with a mean difference of 0.8 (P<0.0001) and I2, 87% (P<0.00001); at 12 weeks the mean difference was 1.25 (P<0.0001) and I2, 92% (P<0.00001).
Table 8 reviews 3 RCTs of electroacupuncture.37-39 Zheng et al conducted a large study of 650 patients with 4 arms: Shu-Mu, He, and He-Shu-Mu acupuncture sites and mosapride (not available in the United States). The primary outcome assessed was an increase in spontaneous BMs at the end of 4 weeks. There was no significant improvement in this measure at 4 weeks, but at week 8, there was a significant mean change of 2.4 in the He group, 2.3 in the Shu-Mu group, and 2.1 in the He-Shu-Mu group compared with 1.4 in the mosapride group (P=0.005).37 In another study, Wu et al compared mosapride with low- and high-current intensity acupuncture at the Qu chí and Shàng jù xu sites and found no significant differences between the groups on primary and secondary outcomes.38 Lastly, in one of the most robust studies, Liu et al evaluated 1,075 patients from 15 centers, comparing electroacupuncture with sham electroacupuncture and found significant improvements in primary outcome. The change in CSBMs from baseline between electro- and sham acupuncture was 0.9 at the end of 8 weeks of treatment (P<0.0001). For secondary outcomes, the mean difference was marginal: 0.24, –0.31, and –0.24 for stool consistency, PAC-QOL score, and straining, respectively (P<0.0001).39
| Table 8. RCTs Assessing Acupuncture Against Sham For Patients With Constipation and Defecation Disorders | ||||
| Study | Patient groups | Intervention dose/duration | Primary end point | Results |
|---|---|---|---|---|
| Zheng et al 2018 | 684 patients with FC (Rome III), 172 He, 171 Shu-Mu, 171 He-Shu-Mu, and 170 mosapride | 16 sessions of acupuncture over 4 wk | Mean weekly SBMs at 4 wk | Mean changes of SBMs at 4 wk were 2.7, 2.7, 2.2, and 2.4 times/wk in He, Shu-Mu, He-Shu-Mu, and mosapride groups, respectively (P=0.226) |
| Wu et al 2017 | 190 patients with FC (Rome III); 58 LCI, 65 HCI, and 67 mosapride | 16 sessions of acupuncture over 4 wk | =3 SBMs/wk and an increase of =1 SBMs/wk from baseline for 3- to 4-wk treatment period | 53.5%, 66.2%, and 52.2% of the patients in LCI, HCI, and mosapride groups reached primary outcome (P>0.05) |
| Liu et al 2016 | 1,075 FC (Rome III); 536 in electroacupuncture, 536 in sham | 28 sessions over 8 wk | Change in mean CSBMs/wk from baseline | Change in mean CSBMs/wk was 1.76 (95% CI, 1.61-1.89) in the electroacupuncture group and 0.87 (95% CI, 0.73-0.97) in the sham group (0.90; 95% CI, 0.74-1.10; P<0.001) |
| CSBM, complete spontaneous bowel movement; FC, functional constipation; HCI, high current intensity; LCI, low current intensity; RCT, randomized controlled trial; SBM, spontaneous BM. Based on references 37-39. | ||||
Treatment for Dyssynergic Defecation
Constipation can be due to multiple etiologies; therefore, different treatment options are needed to target individual pathophysiologic mechanisms. For patients with dyssynergic defecation who are unable to perform coordinated movements of the rectum, puborectalis, and anal sphincter to facilitate stooling, biofeedback therapy has been shown to be an effective treatment.40,41 The American Neurogastroenterology and Motility Society and the European Society of Neurogastroenterology and Motility have given this treatment a Grade A recommendation.42 Biofeedback therapy involves using specialized equipment to measure various physiologic parameters related to bowel muscle function, such as rectal and anal sphincter muscle pressures during attempted defecation. Through biofeedback sessions, patients can learn how to regain voluntary control over their BMs and improve muscle coordination, leading to more effective, coordinated emptying of their bowels.40-43
Home Biofeedback Therapy
Although biofeedback therapy is safe and efficacious, it can be difficult to access medical centers providing biofeedback therapy and skilled personnel to perform traditional office-based biofeedback therapy. Home biofeedback has been developed, in which patients are given the tools and guidance to administer biofeedback treatment from the comfort of their home.43 The advantages of home biofeedback therapy are numerous. It is a cost-effective method for patients to receive readily available treatment, bypassing the difficulty of logistical issues such as transportation and time off work. In addition, it allows patients to access a greater number of treatments according to their own schedules.40 Particularly in cases in which office biofeedback may not be a possibility due to lack of infrastructure, home biofeedback can provide a viable alternative.
In an RCT of 100 patients, Rao et al showed that home biofeedback therapy is effective and safe and was noninferior to office biofeedback therapy.43 Using a rigorous objective outcome measure composed of normalization of dyssynergia pattern plus an increase in 1 or more CSBMs per week, they showed that 68% of patients who received home biofeedback met the primary outcome compared with 70% who received office biofeedback. Satisfaction with bowel function and balloon expulsion time also improved significantly in both groups. In addition, home biofeedback was more cost-effective than office biofeedback and improved QOL domains. However, there is no standardized equipment or protocol or commercial equipment for performing home or office biofeedback therapy. This has handicapped the delivery of biofeedback therapy to patients with pelvic floor disorders. Clearly there is an urgent need for development of innovative biofeedback instruments, and there is early promise.44
Treatment for Rectal Sensory Disorders
Recent studies have highlighted that disorders of rectal sensation play an important role in the pathogenesis of anorectal disorders,45,46 including rectal hyposensitivity, in which there is poor awareness of stooling sensation,45,47 and rectal hypersensitivity, which leads to urgency to defecate even in the presence of a small amount of stool or abdominal pain and is often associated with IBS.48,49
Two recent innovative treatments have been tested in RCTs. In one study, barostat-assisted sensory training (BAST) was compared with syringe-assisted sensory training in patients with rectal hyposensitivity.48 This showed that BAST was superior, with a higher responder rate and greater ease of treatment administration.48 In another study, a novel approach termed sensory adaptation therapy, which involves placing a highly compliant barostat balloon in the rectum and performing gentle, stepwise balloon inflations, led to greater improvement in rectal hypersensitivity and bowel symptoms and was better tolerated than the selective serotonin inhibitor escitalopram.49 Further studies are needed to confirm the beneficial effects of these treatments and other approaches to treat these disorders.
Conclusion
As research and medical advancements continue to expand our understanding of gastrointestinal health, emerging treatments for constipation offer new hope for individuals struggling with this condition. With options from kiwifruit to vibrating pills as well as nonpharmacologic approaches such as biofeedback and neuromodulation therapies, the future looks bright for more effective and personalized treatment options for constipation. However, it is essential to remember that any new treatment should be discussed with qualified healthcare professionals because individual responses to therapies can vary. With ongoing research and continued commitment to improving bowel health, we can look forward to a future where constipation becomes a more manageable and less disruptive problem.
References
- Higgins PDR, Johanson JF. Epidemiology of constipation in North America: a systematic review. Gastroenterology. 2004;99(4):750-759.
- Rao SSC, Brenner DM. Efficacy and safety of over-the-counter therapies for chronic constipation: an updated systematic review. Gastroenterology. 2021;116(6):1156-1181.
- Hosseinzadeh ST, Poorsaadati S, Radkani B, et al. Psychological disorders in patients with chronic constipation. Gastroenterol Hepatol Bed Bench. 2011;4(3):159-163.
- Drossman DA, Hasler WL. Rome IV—functional GI disorders: disorders of gut-brain interaction. Gastroenterology. 2016;150(6):1257-1261.
- Rao SSC, Lacy BE, Emmanuel A, et al. Recognizing and defining occasional constipation: expert consensus recommendations. Am J Gastroenterol. 2022;117(11):1753-1758.
- Gearry R, Fukudo S, Barbara G, et al. Consumption of 2 green kiwifruits daily improves constipation and abdominal comfort-results of an international multicenter randomized controlled trial. Am J Gastroenterol. 2023;118(6):1058-1068.
- Bayer SB, Heenan P, Frampton C, et al. Two gold kiwifruit daily for effective treatment of constipation in adults—a randomized clinical trial. Nutrients. 2022;14(19):4146.
- Chey SW, Chey WD, Jackson K, et al. Exploratory comparative effectiveness trial of green kiwifruit, psyllium, or prunes in US patients with chronic constipation. Am J Gastroenterol. 2021;116(6):1304-1312.
- Rao SSC, Lembo A, Chey WD, et al. Effects of the vibrating capsule on colonic circadian rhythm and bowel symptoms in chronic idiopathic constipation. Neurogastroenterol Motil. 2020;32(11):e13890.
- Mayeli A, Al Zoubi O, White EJ, et al. Parieto-occipital ERP indicators of gut mechanosensation in humans. Nat Commun. 2023;14(1):3398.
- Rao SSC, Quigley EMM, Chey WD, et al. Randomized placebo-controlled phase 3 trial of vibrating capsule for chronic constipation. Gastroenterology. 2023;164(7):1202-1210.
- Zhu JH, Qian YY, Pan J, et al. Efficacy and safety of vibrating capsule for functional constipation (VICONS): a randomised, double-blind, placebo-controlled, multicenter trial. EClinicalMedicine. 2022;47:101407.
- Vitton V, Mion F, Leroi AM, et al. Interferential therapy for chronic constipation in adults: the CON-COUR randomized controlled trial. United European Gastroenterol J. 2023;11(4):337-349.
- Moore JS, Gibson PR, Burgell RE. Randomised clinical trial: transabdominal interferential electrical stimulation vs sham stimulation in women with functional constipation. Aliment Pharmacol Ther. 2020;51(8):760-769.
- Queralto M, Vitton V, Bouvier M, et al. Interferential therapy: a new treatment for slow transit constipation. A pilot study in adults. Colorectal Dis. 2013;15(1):e35-e39.
- Velasco-Benitez C, Villamarin E, Mendez M, et al. Efficacy of transcutaneous posterior tibial nerve stimulation in functional constipation. Eur J Pediatr. 2023;182(3):1309-1315.
- Iqbal F, Collins B, Thomas GP, et al. Bilateral transcutaneous tibial nerve stimulation for chronic constipation. Colorectal Dis. 2016;18(2):173-178.
- Collins B, Norton C, Maeda Y. Percutaneous tibial nerve stimulation for slow transit constipation: a pilot study. Colorectal Dis. 2012;14(4):e165-e170.
- Kumar L, Liwanag J, Athanasakos E, et al. Effectiveness of percutaneous tibial nerve stimulation in managing refractory constipation. Colorectal Dis. 2017;19(1):45-49.
- Gokce AH, Gokce FS, Iliaz R, et al. Transcutaneous tibial nerve stimulation as therapy for functional constipation. Turk J Gastroenterol. 2022;33(7):565-569.
- Gokce AH, Gokce FS. Effects of bilateral transcutaneous tibial nerve stimulation on constipation severity in geriatric patients: a prospective clinical study. Geriatr Gerontol Int. 2020;20(2):101-105.
- Madbouly KM, Abbas KS, Emanuel E. Bilateral posterior tibial nerve stimulation in the treatment of rectal evacuation disorder: a preliminary report. Dis Colon Rectum. 2017;60(3):311-317.
- Zhang N, Huang Z, Xu F, et al. Transcutaneous neuromodulation at posterior tibial nerve and ST36 for chronic constipation. Evid Based Complement Alternat Med. 2014;2014:560802.
- Knowles CH, Horrocks EJ, Bremner SA, et al. Percutaneous tibial nerve stimulation versus sham electrical stimulation for the treatment of faecal incontinence in adults (CONFIDeNT): a double-blind, multicentre, pragmatic, parallel-group, randomised controlled trial. Lancet. 2015;386(10004):1640-1648.
- Jarrett MED, Mowatt G, Glazener CMA, et al. Systematic review of sacral nerve stimulation for faecal incontinence and constipation. Br J Surg. 2004;91(12):1559-1569.
- Kamm MA, Dudding TC, Melenhorst J, et al. Sacral nerve stimulation for intractable constipation. Gut. 2010;59(3):333-340.
- Thomas GP, Duelund-Jakobsen J, Dudding TC, et al. A double-blinded randomized multicentre study to investigate the effect of changes in stimulation parameters on sacral nerve stimulation for constipation. Colorectal Dis. 2015;17(11):990-995.
- Dinning PG, Hunt L, Patton V, et al. Treatment efficacy of sacral nerve stimulation in slow transit constipation: a two-phase, double-blind randomized controlled crossover study. Am J Gastroenterol. 2015;110(5):733-740.
- Zerbib F, Siproudhis L, Lehur PA, et al. Randomized clinical trial of sacral nerve stimulation for refractory constipation. Br J Surg. 2017;104(3):205-213.
- Kim JS, Yi SJ. Effects of low-frequency current sacral dermatome stimulation on idiopathic slow transit constipation. J Phys Ther Sci. 2014;26(6):831-832.
- Iqbal F, Thomas GP, Tan E, et al. Transcutaneous sacral electrical stimulation for chronic functional constipation. Dis Colon Rectum. 2016;59(2):132-139.
- Yang Y, Yim J, Choi W, et al. Improving slow-transit constipation with transcutaneous electrical stimulation in women: a randomized, comparative study. Women Health. 2017;57(4):494-507.
- McClurg D, Booth L, Herrero-Fresneda I. Safety and efficacy of intermittent colonic exoperistalsis device to treat chronic constipation: a prospective multicentric clinical trial. Clin Transl Gastroenterol. 2020;11(12):e00267.
- Yao JP, Chen LP, Xiao XJ, et al. Effectiveness and safety of acupuncture for treating functional constipation: an overview of systematic reviews. J Integr Med. 2022;20(1):13-25.
- Jie L, Shiping L, Yue X, et al. Efficacy and safety of electroacupuncture for secondary constipation: a systematic review and meta-analysis. Int J Colorectal Dis. 2023;38(1):196.
- Zhang N, Hou L, Yan P, et al. Electro-acupuncture vs. sham electro-acupuncture for chronic severe functional constipation: a systematic review and meta-analysis. Complement Ther Med. 2020;54:102521.
- Zheng H, Liu ZS, Zhang W, et al. Acupuncture for patients with chronic functional constipation: a randomized controlled trial. Neurogastroenterol Motil. 2018;30(7):e13307.
- Wu X, Zheng C, Xu X, et al. Electroacupuncture for functional constipation: a multicenter, randomized, control trial. Evid Based Complement Alternat Med. 2017;2017:1428943.
- Liu Z, Yan S, Wu J, et al. Acupuncture for chronic severe functional constipation: a randomized trial. Ann Intern Med. 2016;165(11):761-769.
- Rao SSC, Go JT, Valestin J, et al. Home biofeedback for the treatment of dyssynergic defecation: does it improve quality of life and is it cost-effective? Am J Gastroenterol. 2019;114(6):938-944.
- Rao SS. Biofeedback therapy for constipation in adults. Best Pract Res Clin Gastroenterol. 2011;25(1):159-166.
- Rao SS, Benninga MA, Bharucha AE, et al. ANMS-ESNM position paper and consensus guidelines on biofeedback therapy for anorectal disorders. Neurogastroenterol Motil. 2015;27(5):594-609.
- Rao SSC, Valestin JA, Xiang X, et al. Home-based versus office-based biofeedback therapy for constipation with dyssynergic defecation: a randomised controlled trial. Lancet Gastroenterol Hepatol. 2018;3(11):768-777.
- Zhou J, Ho V, Javadi B. New internet of medical things for home-based treatment of anorectal disorders. Sensors (Basel). 2022;22(2):625.
- Rao SS, Bharucha AE, Chiarioni G, et al. Functional anorectal disorders. Gastroenterology. Published online March 25, 2016. doi:10.1053/j.gastro.2016.02.009
- Vollebregt PF, Burgell RE, Hooper RL, et al. Clinical impact of rectal hyposensitivity: a cross-sectional study of 2,876 patients with refractory functional constipation. Am J Gastroenterol. 2021;116(4):758-768.
- Chang L. The role of stress on physiologic responses and clinical symptoms in irritable bowel syndrome. Gastroenterology. 2011;140(3):761-765.
- Rao SSC, Yan Y, Erdogan A, et al. Barostat or syringe-assisted sensory biofeedback training for constipation with rectal hyposensitivity: a randomized controlled trial. Neurogastroenterol Motil. 2022;34(3):e14226.
- Rao SSC, Coss-Adame E, Yan Y, et al. Sensory adaptation training or escitalopram for IBS with constipation and rectal hypersensitivity: a randomized controlled trial. Clin Transl Gastroenterol. 2021;12(7):e00381.
Copyright © 2024 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.
Download to read this article in PDF document:![]() Emerging Treatments for Chronic Constipation And Defecation Disorders
Emerging Treatments for Chronic Constipation And Defecation Disorders