Professor, Department of Medicine
University of Louisville
Louisville, Kentucky
Alcohol-associated liver disease, one of the most common types of liver disease worldwide, is associated with tremendous morbidity, mortality, and financial costs.1,2 Although significant progress has been made in screening and management of patients with early, or nonadvanced, ALD, there remain unmet clinical needs and areas of future prospects in the management.2
Spectrum and Definitions of ALD
ALD is caused by alcohol use disorder (AUD)—harmful alcohol consumption (=2 drinks per day or >7 per week in females and =3 drinks daily or >14 weekly in males) despite negative physical, social, and interpersonal consequences accompanied by dependence, withdrawal, and tolerance of alcohol.1
Manifestation of ALD (Figure 1) varies from nonadvanced disease (steatosis with or without steatohepatitis and fibrosis stage =2 or periportal fibrosis) to advanced disease (advanced or stage 3 fibrosis and cirrhosis).1 Steatosis is defined as more than 5% of hepatocytes containing fat droplets.3 Ultrasound examination accurately identifies patients with fat droplets in more than 33% of hepatocytes (moderate or severe steatosis).1 Controlled attenuation parameter and magnetic resonance–proton density fat fraction can detect mild steatosis, but high cost and lack of availability—as the technology is often only at tertiary centers—limit their widespread use.1 Steatohepatitis is identified on liver histology with features such as neutrophilic lobular infiltration, hepatocyte ballooning, Mallory hyaline, and steatosis.1

Along with morbidity and mortality, the economic burden—defined as direct costs of taking care of patients and indirect costs from lost disease-adjusted life-years—is mainly due to complications and requirement for liver transplantation in patients with advanced disease.2 There were 123 million individuals worldwide and 2.2 million in the United States with ALD cirrhosis in 2017.2 The highest ALD burden is in Europe, but the magnitude of ALD is increasing in Asia,2 sub-Sahara Africa,2 particularly in young people aged 25 to 34 years,4 and in American Indians.5-7
Compared with other liver diseases, ALD more often presents at an advanced stage.1,8 Opportunities for detecting ALD and intervening early to prevent advanced disease often are missed during clinical encounters with healthcare providers.8,9 Clearly, screening for alcohol use at every healthcare encounter is important to identify those at risk for ALD, and to determine the prevalence of nonadvanced disease.8,10
Screening for Alcohol Use
Screening for alcohol history provides an opportunity to 1) administer brief counseling sessions for occasional heavy drinkers; 2) refer to addiction treatment for regular harmful users; 3) evaluate for end-organ damage; and 4) evaluate for psychiatric problems (anxiety, depression, insomnia, or other substance use disorder) and social and interpersonal issues (job performance, marital and domestic issues).10,11 Brief counseling sessions reduce patient alcohol consumption by helping to improve physical and mental health, and in turn, decrease hospital visits and motor vehicle accidents.10,11
The Alcohol Use Disorder Identification Test (AUDIT) is a 10-item questionnaire tool, with each answer self-reported by the patient on a scale from 0 to 4.1,10-13 A brief version of AUDIT, which consists of the first 3 questions (AUDIT-C), is a simplified version that efficiently helps to identify heavy drinkers with excellent reliability and validity (total score of >4 in female and >6 in male patients).10-13
Together with this screening tool, indirect alcohol biomarkers are increasingly being used because patients may not provide accurate information due to the stigma surrounding drinking behaviors and AUD. The presence of 2 or more DSM-5 criteria defines AUD, with severity increasing with the number of criteria in a given individual.11,12 The alcohol metabolites ethyl-glucuronide (eTG) and phosphatidylethanol (PEth) can be used for identifying alcohol use.11,12,14 Furthermore, eTG can detect alcohol consumption within 2 to 3 days and PEth within 2 to 4 weeks from last use of alcohol.11,12 Aside from a longer window of detection, PEth does not result in false-positive tests, levels correlate with the amount of consumption, and it allows follow-up over time in individuals undergoing treatment.11
Screening for Liver Disease
Liver biochemical blood tests and liver ultrasound are recommended to screen for ALD in people with harmful alcohol use.1,11,12 Among those with evidence of liver disease, detected by transaminitis, imaging abnormalities, or both, other causes of liver disease should be ruled out before diagnosing ALD (Table 1).1 The Alcohol-associated steatohepatitis–nonalcoholic steatohepatitis index can differentiate ALD and metabolic-associated steatotic liver disease (formerly known as nonalcoholic fatty liver disease) in obese individuals with harmful alcohol use.11,15
| Table 1. Criteria for Diagnosis of Alcohol-Associated Liver Disease |
| Harmful alcohol use (>2 drinks/d in females and >3 drinks/d in males) for =12 mo |
| Liver disease diagnosed in the presence of clinical signs of cirrhosis, biochemical liver abnormalities, or liver imaging findings (steatotic liver disease [formerly fatty liver], portal hypertension, cirrhosis) |
| When liver aminotransferases are elevated, they are usually <400 IU/L with AST:ALT ratio >1. |
Exclude other causes of liver disease:
|
| ALT, alanine aminotransferase; AST, aspartate aminotransferase; IU, international unit. Based on reference 1. |
Assessment of Fibrosis Using Noninvasive Tests
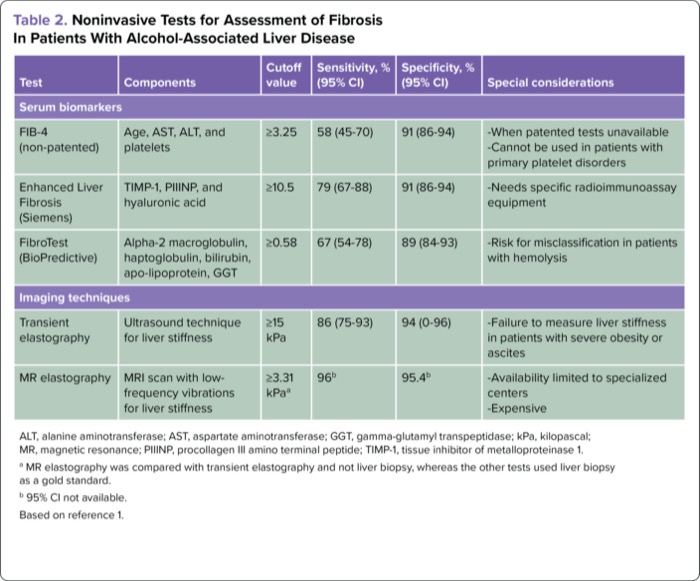
Liver biopsy is the gold-standard test for determining the fibrosis stage and diagnosing nonadvanced ALD.12 Given the limitations of liver biopsy due to invasiveness with a potential for complications, cost, and sampling error in up to 20% of cases, noninvasive tests—serological and/or radiological tests—are gaining acceptance for determining the fibrosis risk and stratifying the disease spectrum in most liver disease, including ALD.11,12 Of the serological tests, non-patented tests that do not require additional labs other than those obtained for routine patient care, such as the Fibrosis-4 (FIB-4) scoring system, are preferred.1 At a cutoff value of 3.25, FIB-4 has a specificity of 90%, but a lower sensitivity of 60%.1 FibroTest (BioPredictive) and the Enhanced Liver Fibrosis (ELF) test (Siemens) are patented tests that require additional labs, but have a sensitivity of 70% to 80% at cutoff values of 0.58 and 10.5, respectively (Table 2).1,12
| Table 2. Noninvasive Tests for Assessment of Fibrosis In Patients With Alcohol-Associated Liver Disease | |||||
| Test | Components | Cutoff value | Sensitivity, % (95% CI) | Specificity, % (95% CI) | Special considerations |
|---|---|---|---|---|---|
| Serum biomarkers | |||||
| FIB-4 (non-patented) | Age, AST, ALT, and platelets | =3.25 | 58 (45-70) | 91 (86-94) |
|
| Enhanced Liver Fibrosis (Siemens) | TIMP-1, PIIINP, and hyaluronic acid | =10.5 | 79 (67-88) | 91 (86-94) |
|
| FibroTest (BioPredictive) | Alpha-2 macroglobulin, haptoglobulin, bilirubin, apo-lipoprotein, GGT | =0.58 | 67 (54-78) | 89 (84-93) |
|
| Imaging techniques | |||||
| Transient elastography | Ultrasound technique for liver stiffness | =15 kPa | 86 (75-93) | 94 (0-96) |
|
| MR elastography | MRI scan with low--frequency vibrations for liver stiffness | =3.31 kPaa | 96b | 95.4b |
|
| ALT, alanine aminotransferase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transpeptidase; kPa, kilopascal; MR, magnetic resonance; PIIINP, procollagen III amino terminal peptide; TIMP-1, tissue inhibitor of metalloproteinase 1. a MR elastography was compared with transient elastography and not liver biopsy, whereas the other tests used liver biopsy as a gold standard. b 95% CI not available. Based on reference 1. | |||||
Of the radiological tests, transient elastography (TE), commonly FibroScan (Echosens), is a validated and point-of-care test and is widely available. With ultrasound-based technique, TE measures liver stiffness as the velocity of displaced liver tissue by the shear waves sent through the liver via the probe on the patient’s skin, (reported in kilopascals).1,12 Liver inflammation and/or elevated aspartate aminotransferase and bilirubin in active alcohol users may overestimate liver stiffness, and repeat TE after the patient has abstained from alcohol for at least 2 weeks is recommended.1,12 The pooled data on 1,026 patients from 10 studies assessing TE in ALD patients showed that a cutoff value of liver stiffness less than 12 excluded advanced disease with an accuracy of 83% to 91%.16
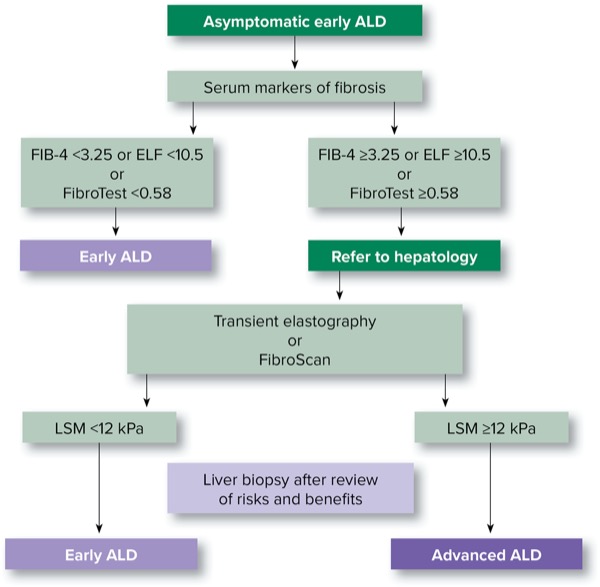
Transient elastography is limited by its inability to assess the hepatic parenchyma and technical issues in obese individuals. Magnetic resonance elastography is more accurate, but it is limited by high cost, lack of data in ALD patients, and lack of accessibility because it is available mostly at specialized centers or in research units.1,12 In routine clinical practice, FIB-4 followed by TE is recommended if there is a suspicion of advanced ALD (Figure 2).1
Treatment of Nonadvanced ALD
With the lack of pharmacologic therapies, treatment of nonadvanced ALD rests on risk factor control of alcohol use to prevent progression to cirrhosis and advanced–stage disease. Achieving complete abstinence is the goal, but this may be difficult for the majority of patients. Specific therapies targeted at AUD include behavioral and pharmacologic interventions.
Behavioral Therapies for AUD
Cognitive behavioral therapy and motivational enhancement therapy aim to identify triggers resulting in relapse.17 Motivational enhancement focuses on stimulating the patient’s motivation for change. The focus of cognitive therapy is to identify triggers that lead to alcohol use and develop coping skills to prevent relapse. There is a lack of data on use of these therapies in patients with nonadvanced ALD.
Pharmacologic Therapies for AUD
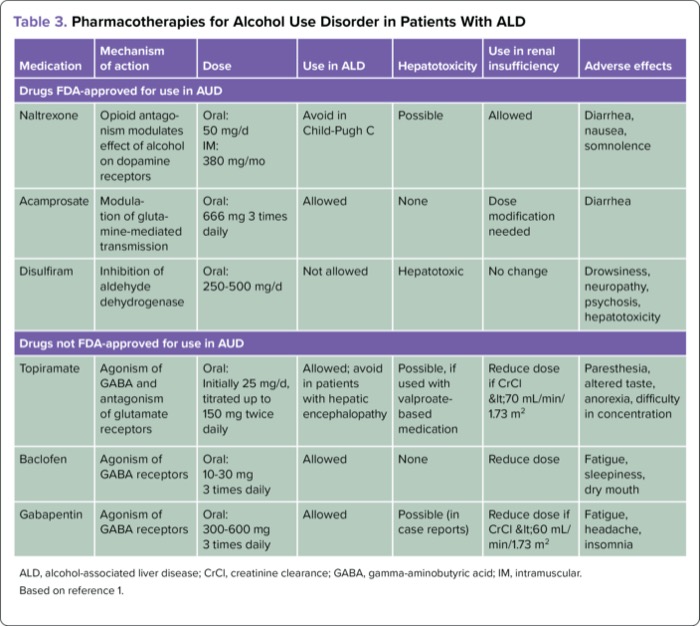
Data on the efficacy and safety of various drugs for AUD treatment specifically in patients with ALD are scant. Of the FDA-approved drugs, disulfiram is completely metabolized in the liver and is contraindicated in ALD patients. Naltrexone, an opioid receptor antagonist, and acamprosate, which works as an antagonist to glutaminergic activity and promotes gamma-aminobutyric receptor agonism, are safe in patients with nonadvanced ALD.1,12,13 Of the several drugs not approved for AUD, baclofen, gabapentin, and topiramate have data on their use: Patients with kidney disease should avoid acamprosate and topiramate at a creatinine clearance less than 30 mL per minute, and their dose should be reduced to between 30 and 60 mL per minute (Table 3).1,12
| Table 3. Pharmacotherapies for Alcohol Use Disorder in Patients With ALD | ||||||
| Medication | Mechanism of action | Dose | Use in ALD | Hepatotoxicity | Use in renal insufficiency | Adverse effects |
|---|---|---|---|---|---|---|
| Drugs FDA-approved for use in AUD | ||||||
| Naltrexone | Opioid antagonism modulates effect of alcohol on dopamine receptors | Oral: 50 mg/d IM: 380 mg/mo | Avoid in Child-Pugh C | Possible | Allowed | Diarrhea, nausea, somnolence |
| Acamprosate | Modulation of glutamine-mediated transmission | Oral: 666 mg 3 times daily | Allowed | None | Dose modification needed | Diarrhea |
| Disulfiram | Inhibition of aldehyde dehydrogenase | Oral: 250-500 mg/d | Not allowed | Hepatotoxic | No change | Drowsiness, neuropathy, psychosis, hepatotoxicity |
| Drugs not FDA-approved for use in AUD | ||||||
| Topiramate | Agonism of GABA and antagonism of glutamate receptors | Oral: Initially 25 mg/d, titrated up to 150 mg twice daily | Allowed; avoid in patients with hepatic encephalopathy | Possible, if used with valproate-based medication | Reduce dose if CrCl <70 mL/min/1.73 m2 | Paresthesia, altered taste, anorexia, difficulty in concentration |
| Baclofen | Agonism of GABA receptors | Oral: 10-30 mg 3 times daily | Allowed | None | Reduce dose | Fatigue, sleepiness, dry mouth |
| Gabapentin | Agonism of GABA receptors | Oral: 300-600 mg 3 times daily | Allowed | Possible (in case reports) | Reduce dose if CrCl <60 mL/min/1.73 m2 | Fatigue, headache, insomnia |
| ALD, alcohol-associated liver disease; CrCl, creatinine clearance; GABA, gamma-aminobutyric acid; IM, intramuscular. Based on reference 1. | ||||||
Prevention of ALD
Alcohol consumption is the third most important preventable cause of any disease after smoking and hypertension. Prevention is always better than cure for any disease, including ALD. This can be achieved by eliminating the exposure to alcohol (primary prevention) or AUD treatment for people with nonadvanced ALD (secondary prevention). Of several regulatory strategies, control of a minimum unit price of alcohol, ban on alcohol advertisements, and restriction on availability of alcohol are the most effective for reducing alcohol use at the mass level, which directly translate to improved population liver health.1,12 However, the strong lobby of manufacturers and distributors of alcohol is a major barrier to implementing these strategies.
Conclusion
ALD is a leading cause of liver-related morbidity and mortality. Although significant progress has been made in screening and management, there remain unmet clinical needs and areas of future prospects in the management of patients with nonadvanced ALD (Table 4). More research is needed to find new pharmacotherapies and biomarkers for assessing risk factors and disease progression. In addition, policy changes could help drive population-level prevention efforts.
| Table 4. Clinical Unmet Needs In Early Nonadvanced ALD |
| Prevalence and magnitude |
| Noninvasive biomarkers for accurate assessment of fibrosis and steatohepatitis |
| Animal model mimicking human phenotype of ALD and AH |
| Newer therapeutic targets and pharmacotherapies |
| End points to determine safe alcohol use limit |
| Accurate cost-effective biomarkers for monitoring of alcohol consumption |
| Clinical trials to determine safety of drugs to treat alcohol use disorder |
| Funding and industry collaboration on research |
| National policies and cost-effective measures to reduce alcohol consumption |
| International collaboration with global understanding of and treatments for ALD |
| AH, alcoholic hepatitis; ALD, alcohol-associated liver disease. Based on reference 11. |
References
- Singal AK, Mathurin P. Diagnosis and management of alcohol-associated liver disease: a review. JAMA. 2021;326(2):165-176.
- GBD 2017 Cirrhosis Collaborators. The global, regional, and national burden of cirrhosis by cause in 195 countries and territories, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol Hepatol. 2020;5(3):245-266.
- Nassir F, Rector RS, Hammoud GM, et al. Pathogenesis and prevention of hepatic steatosis. Gastroenterol Hepatol (NY). 2015;11(3):167-175.
- Singal AK, Arora S, Wong RJ, et al. Increasing burden of acute-on-chronic liver failure among alcohol-associated liver disease in the young population in the United States. Am J Gastroenterol. 2020;115(1):88-95.
- Kulkarni NS, Wadhwa DK, Kanwal F, et al. Alcohol-associated liver disease mortality rates by race before and during the COVID-19 pandemic in the US. JAMA Health Forum. 2023;4(4):e230527.
- Kardashian A, Serper M, Terrault N, et al. Health disparities in chronic liver disease. Hepatology. 2023;77(4):1382-1403.
- Suryaprasad A, Byrd KK, Redd JT, et al. Mortality caused by chronic liver disease among American Indians and Alaska Natives in the United States, 1999-2009. Am J Public Health. 2014;104 Suppl 3(Suppl 3):S350-S358.
- Shah ND, Ventura-Cots M, Abraldes JG, et al. Alcohol-related liver disease is rarely detected at early stages compared with liver diseases of other etiologies worldwide. Clin Gastroenterol Hepatol. 2019;17(11):2320-2329.e12.
- Askgaard G, Leon DA, Kjaer MS, et al. Risk for alcoholic liver cirrhosis after an initial hospital contact with alcohol problems: a nationwide prospective cohort study. Hepatology. 2017;65(3):929-937.
- O’Connor EA, Perdue LA, Senger CA, et al. Screening and behavioral counseling interventions to reduce unhealthy alcohol use in adolescents and adults: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2018;320(18):1910-1928.
- Singal AK, Bataller R, Ahn J, et al. ACG clinical guideline: alcoholic liver disease. Am J Gastroenterol. 2018;113(2):175-194.
- European Association for the Study of the Liver. EASL clinical practice guidelines: Management of alcohol-related liver disease. J Hepatol. 2018;69(1):154-181.
- Kranzler HR, Soyka M. Diagnosis and pharmacotherapy of alcohol use disorder: a review. JAMA. 2018;320(8):815-824.
- Perilli M, Toselli F, Franceschetto L, et al. Phosphatidylethanol (PEth) in Blood as a Marker of Unhealthy Alcohol Use: A Systematic Review with Novel Molecular Insights. Int J Mol Sci. 2023;24(15):12175.
- Cerovic I, Mladenovic D, Ješic R, et al. Alcoholic liver disease/nonalcoholic fatty liver disease index: distinguishing alcoholic from nonalcoholic fatty liver disease. Eur J Gastroenterol Hepatol. 2013;25(8):899-904. doi:10.1097/MEG.0b013e32835f0786.
- Nguyen-Khac E, Thiele M, Voican C, et al. Non-invasive diagnosis of liver fibrosis in patients with alcohol-related liver disease by transient elastography: an individual patient data meta-analysis. Lancet Gastroenterol Hepatol. 2018;3(9):614-625.
- Ray LA, Meredith LR, Kiluk BD, et al. Combined pharmacotherapy and cognitive behavioral therapy for adults with alcohol or substance use disorders: a systematic review and meta-analysis. JAMA Netw Open. 2020;3(6):e208279.
Copyright © 2023 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.
Download to read this article in PDF document:![]() Screening, Diagnosis, and Treatment of Nonadvanced Alcohol-Associated Liver Disease
Screening, Diagnosis, and Treatment of Nonadvanced Alcohol-Associated Liver Disease