Originally published by our sister publication Pharmacy Practice News
Visantea
St. Paul MN
Visante
St. Paul, MN
A series of events could lead up to a supply chain disruption of one of our most critical medical devices. On November 30, 2023, the FDA issued a safety communication alerting consumers, healthcare providers, and medical facilities regarding an ongoing evaluation of potential device malfunctions linked to syringes manufactured in China and sold under familiar American names.1
The FDA has received reports detailing quality concerns related to multiple Chinese syringe manufacturers. Issues reported include leaks, breakage, and additional issues due to manufacturer changes made to the syringe dimensions. These quality issues may affect the performance and safety of the syringes, including their ability to deliver the correct dose of medication when used alone or with other medical devices such as infusion pumps.
The agency is expressing apprehension that specific syringes originating from China might lack uniformity and fail to meet the required standards of quality and performance. Currently glass syringes, oral syringes, and syringes manufactured in the United States are not affected by this communication.
Of note, a majority of US brands source their syringes from Chinese manufacturers. At this time Becton, Dickinson and Company (BD) is the only domestic syringe manufacturer that sells under its own label. The current recalls cover more than 45 million disposable syringes in the US market. In the November 30, 2023, communication, the FDA stated it will continue to monitor for additional quality and safety concerns and may prevent further shipments of Chinese-made syringes from entering the United States. The affected supply of syringes, in addition to continued diligence by the FDA to monitor ongoing supplies from China, may lead to shortages of certain sizes to all syringes and increase in costs.
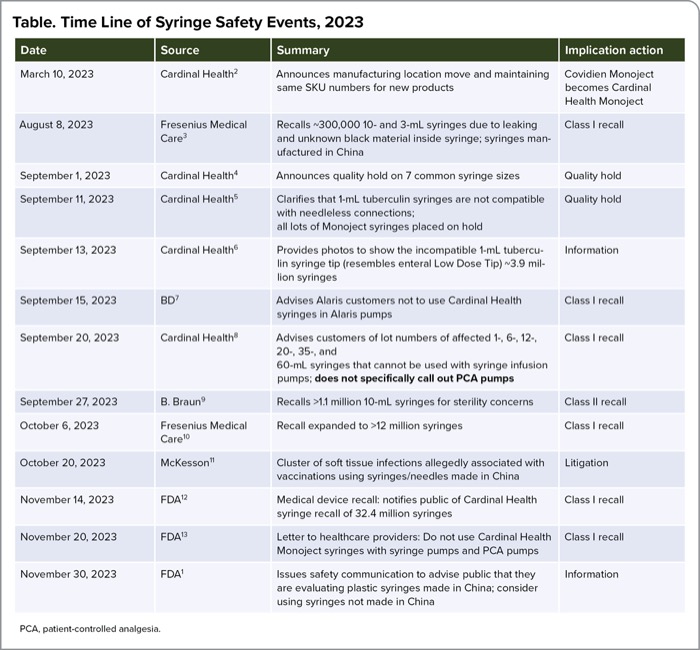
Before the FDA’s communication, US medical device manufacturers of syringes had cautioned about compatibility problems between their syringe with syringe infusion pumps and other quality problems. The time line in the Table highlights events leading up to the November 30 FDA communication.
Standards for Syringes
Syringes are often viewed as an interchangeable commodity with little to no concern for variations. The FDA “clears” (approves for marketing) syringes for use in the United States under the 510(k) device process. The FDA utilizes a 3-class system to classify medical devices based on risk, where Class I devices (ie, bandages) have the lowest risk to patients and Class III devices represent the highest risk category (ie, pacemakers, implants). Syringes are classified as a Class II medical device. Class II medical devices carry a moderate to high risk to the patient and/or user.
The International Organization for Standardization (ISO) sets global standards for syringes, ensuring their quality, safety, and performance. ISO has established essential standards for syringes:
ISO 7886-1:2017 Sterile hypodermic syringes for single use — Part 1: Syringes for manual use applies to syringes that healthcare professionals manually operate, and ISO 7886-2:2020 Sterile hypodermic syringes for single use Part 2: Syringes for use with power-driven syringe pumps focuses on syringes designed for compatibility with automated medical devices. These guidelines are in place to ensure that syringes meet the following criteria:
- manufactured using tested and approved materials that meet strict safety standards;
- contain accurate graduations for dosing (with allowance for accuracy variances);
- effectively deliver medications;
- maintain sterility; and
- reliably and consistently function as designed.
Within the ISO standards is the allowance and encouragement for innovation. Therefore, they allow for some design variations and accuracy tolerances. This flexibility enables manufacturers to develop new and improved syringes with the expectations of not compromising performance or failing to meet testing requirements. The ISO allowances pose some key limitations that may have contributed to this safety notice such as the following:
- Compliance: While ISO standards set guidelines for manufacturing and quality control, they might not guarantee absolute safety or effectiveness in all situations. As we have experienced, compliance with standards does not guarantee the syringe is immune to defects.
- Scope: ISO standards do not encompass all aspects of syringe usage or address every potential risk associated with their use. Certain specific applications or unique circumstances might not be fully covered by these standards.
- Evolving technology: Rapid advancements in technology and materials have outpaced the standards (ie, syringe pumps, compounding robotics).
- Standards antiquity: Standards may not always reflect the latest best practices. Continuous improvement in manufacturing techniques or safety features might not be immediately integrated into standards.
- Global variations: Standards might not fully align with specific regulations or practices in different countries or regions, leading to variations in compliance and potential interpretation of the standards.
- Change control: Unlike the drug industry, minor changes to devices can happen without retesting and validating that the change has not affected performance and safety.
Manufacturers, healthcare providers, and regulatory bodies must not solely rely on the FDA 510(k) process, ISO standards, and syringe pump manufacturers’ validation processes as guarantees of performance but must also consider additional factors and best practices to ensure the safe and effective use of syringes in various medical applications.
Impact to Healthcare Facilities
From the November 2023 FDA communication, FDA recommendations for consumers, healthcare providers, and facilities:
- The FDA advises sites to inspect their current supplies and check the manufacturing location for syringes by reviewing the labeling, outer packaging, or contacting your supplier or group purchasing organization syringes manufactured in China.
- Consider using syringes not manufactured in China, if possible. At this time, glass syringes, prefilled syringes, or syringes used for oral or topical purposes are not included.
- If you only have syringes manufactured in China, then continue to use them as needed until you are able to use alternative syringes and closely monitor for leaks, breakage, and other problems.
- Report any issues with syringes through the FDA MedWatch online voluntary reporting form (https://www.accessdata.fda.gov/scripts/ medwatch/ index.cfm?action=reporting.home).
In addition to FDA recommendations, healthcare facilities should also consider the following:
- Do not assume that a US brand or distributor name means the syringe is manufactured in the United States. Many syringes are produced by a third party under a private label agreement.
- Inspect all areas for syringes: nurse stations, patient rooms, treatment rooms, operating room suites, infusion centers, home infusion, hospital in the home models, hospice centers, radiology treatment, cardiovascular diagnosis/treatment, physician offices, outreach clinics, and emergency squads as examples.
- Ensure through the inspection process the ability to identify the pedigree of the syringe and its components per the FDA communication by visually inspecting the packaging of syringes or requesting from the manufacturer a pedigree listing of all components of the syringe.
- Ensure syringes used in syringe pumps are compatible in their current form and meet the syringe pump testing standards. If unable to validate a change control and communication system for syringes used in syringe pumps, it cannot be assumed that changes do not affect pump functionality.
- Notify patients who have syringes that are dispensed for home therapies such as patient-controlled analgesia (PCA) pumps and syringe pumps.
- Inspect kits (surgical kits) created internally that may include syringes, and kits provided to organizations by third-party vendors.
- Establish redundant contracting for syringes manufactured in the United States.
Impact to Healthcare Providers
The immediate impact is already being felt by healthcare providers who experience a poorly made syringe. Leaky syringes used during preparation and the administration of drugs is a concern, as noted in the recalls listed in the Table. An additional risk is for hazardous drugs. A leaky conventional syringe places an additional risk to healthcare providers and patients due to direct exposure to hazardous drugs.
| Table. Time Line of Syringe Safety Events, 2023 | |||
| Date | Source | Summary | Implication action |
|---|---|---|---|
| March 10, 2023 | Cardinal Health2 | Announces manufacturing location move and maintaining same SKU numbers for new products | Covidien Monoject becomes Cardinal Health Monoject |
| August 8, 2023 | Fresenius Medical Care3 | Recalls ~300,000 10- and 3-mL syringes due to leaking and unknown black material inside syringe; syringes manufactured in China | Class I recall |
| September 1, 2023 | Cardinal Health4 | Announces quality hold on 7 common syringe sizes | Quality hold |
| September 11, 2023 | Cardinal Health5 | Clarifies that 1-mL tuberculin syringes are not compatible with needleless connections; all lots of Monoject syringes placed on hold | Quality hold |
| September 13, 2023 | Cardinal Health6 | Provides photos to show the incompatible 1-mL tuberculin syringe tip (resembles enteral Low Dose Tip) ~3.9 million syringes | Information |
| September 15, 2023 | BD7 | Advises Alaris customers not to use Cardinal Health syringes in Alaris pumps | Class I recall |
| September 20, 2023 | Cardinal Health8 | Advises customers of lot numbers of affected 1-, 6-, 12-, 20-, 35-, and 60-mL syringes that cannot be used with syringe infusion pumps; does not specifically call out PCA pumps | Class I recall |
| September 27, 2023 | B. Braun9 | Recalls >1.1 million 10-mL syringes for sterility concerns | Class II recall |
| October 6, 2023 | Fresenius Medical Care10 | Recall expanded to >12 million syringes | Class I recall |
| October 20, 2023 | McKesson11 | Cluster of soft tissue infections allegedly associated with vaccinations using syringes/needles made in China | Litigation |
| November 14, 2023 | FDA12 | Medical device recall: notifies public of Cardinal Health syringe recall of 32.4 million syringes | Class I recall |
| November 20, 2023 | FDA13 | Letter to healthcare providers: Do not use Cardinal Health Monoject syringes with syringe pumps and PCA pumps | Class I recall |
| November 30, 2023 | FDA1 | Issues safety communication to advise public that they are evaluating plastic syringes made in China; consider using syringes not made in China | Information |
| PCA, patient-controlled analgesia. | |||
Syringes that vary in calibration and sizes pose additional challenges to healthcare providers, as they relate to dose accuracy. Some syringes have different barrel markings, 0.1 mL versus 0.2 mL, which can lead to dosing errors if syringe brands are switched, or manufacturers of syringes make changes, and the healthcare provider is not apprised of the change.
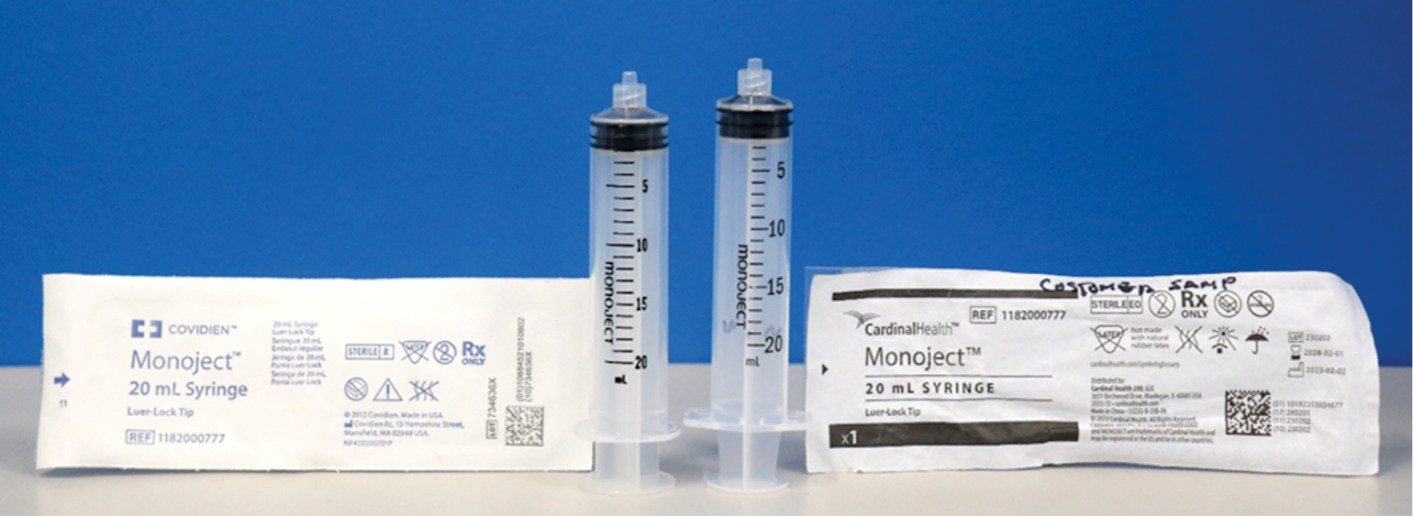
Syringes that vary in physical size but deliver the same quantity create issues with syringe pumps and robotic compounding devices that have been calibrated to a specific size. The dimensional variances may result in syringe pump issues leading to potential underdosing, overdosing, occlusion alarms, and potential delays in therapies. The FDA highlighted the dimensional variances in the November 14, 2023, FDA medical device recall: Cardinal Health recalls Monoject disposable syringes for incompatibilities with syringe pumps.12 The FDA posted a picture of dimensional variances of Monoject 10-mL syringes that deliver the same volume. The Figure shows an example of dimensional differences between 2 syringes, both named “Monoject.” Once the syringe is removed from the package, the distinction between Covidien and Cardinal Health brands is gone. Syringe and PCA pumps have libraries of validated syringes, often labeled simply “Monoject.” This has caused a Class I recall/product correction for Alaris PCA and syringe pump users to update their compatibility lists.14
The current situation may be further complicated if the FDA chooses to temporarily prohibit importation of syringes from China. Hospitals that prepare and administer antibiotics as IV push should consider using a mini-bag system as an alternative to conserve 10-mL syringes. If you purchase medications that are prepared by commercial compounding companies, consult with the company to ensure that they are not using syringes from China, or if they have recently switched away from China syringes, that drug stability testing has been done with the new brand of syringes.
Conclusion
The syringe is one of the most critical medical devices used in medicine. Its impact on healthcare is undeniable for the way medications, vaccines, and other treatments are prepared and administered. Pedigree matters: How and where was the syringe manufactured? Where were the components sourced? What company will stand behind the syringe if there is a problem? Our confidence in the simple disposable syringe depends on quality, consistency, and performance. We cannot view syringes as a commodity today.
References
- FDA. Evaluating plastic syringes made in China for potential device failures: FDA safety communication. November 30, 2023. Accessed December 29, 2023. https://www.fda.gov/medical-devices/ safety-communications/ evaluating-plastic-syringes-made-china-potential-device-failures-fda-safety-communication
- Cardinal Health customer letter. RE: Change notification Cardinal Health product enhancements notifications 03-10-2023. March 24, 2023. Personal files.
- FDA. Fresenius Medical Care recalls Sanxin Single Use Syringes. August 8, 2023. Accessed December 29, 2023. https://www.fda.gov/medical-devices/ medical-device-recalls/ fresenius-medical-care-recalls-sanxin-single-use-syringes-leakages
- Cardinal Health customer letter. RE: Cardinal quality notifications. September 1, 2023. Personal files.
- Cardinal Health customer letter. RE: Cardinal quality notifications. September 11, 2023. Personal files.
- Cardinal Health. Cardinal Health urgent medical device product correction. RE: Cardinal Health? Monoject? 1 mL Tuberculin Syringe Luer-Lock Tip Soft Pack. September 13, 2023. Accessed December 29, 2023. https://www.medline.com/media/ assets/ pdf/ vendor-list/ Cardinal_Health_Monoject_1_ML_Final_Letter_Event_2023_05724_9_13_23.pdf
- BD customer letter. Urgent medical device labeling correction. BD Alaris? Syringe Module, BD Alaris? PCU and Alaris? PCA Module. Type of action: medical device labeling correction. September 15, 2023. Personal file.
- Cardinal Health. Cardinal Health urgent medical device product correction. RE: all lot numbers of syringes not to be used with syringe pumps. September 20, 2023. Accessed December 29, 2023. https://www.medline.com/media/assets/pdf/vendor-list/Cardinal_Health_Monoject_Market_Notification_Letter_Event_2023_05729_9_20_23_(003).pdf
- FDA. Class 2 device recall B. Braun NormJect syringe. September 27, 2023. Accessed December 29, 2023. https://www.accessdata.fda.gov/scripts/ cdrh/ cfdocs/ cfres/ res.cfm?id=202871
- FDA. Fresenius Medical Care recalls Sanxin single use syringes for leakages. Updated number of syringes affected. October 6, 2023. Accessed December 29, 2023. https://www.fda.gov/medical-devices/ medical-device-recalls/ fresenius-medical-care-recalls-sanxin-single-use-syringes-leakages
- Marchante M, Weaver J. Miami kids needed surgery after infections set in. Were contaminated needles to blame? Miami Herald. October 20, 2023. Accessed December 29, 2023. https://www.miamiherald.com/news/ health-care/ article280651660.html
- FDA. Cardinal Health recalls Monoject disposable syringes for incompatibilities with syringe pumps. November 14, 2023. Accessed December 29, 2023. https://www.fda.gov/medical-devices/ medical-device-recalls/ cardinal-health-recalls-monoject-disposable-syringes-incompatibilities-syringe-pumps
- FDA. Do not use Cardinal Health Monoject Syringes with syringe pumps and PCA pumps. November 20, 2023. Accessed December 29, 2023. https://www.fda.gov/medical-devices/ letters-health-care-providers/ do-not-use-cardinal-health-monoject-syringes-syringe-pumps-and-pca-pumps#other
- FDA. Becton Dickinson (BD)/Carefusion 303 recalls Alaris Infusion Pumps due to compatibility issues with Cardinal Health Monoject Syringes. December 1, 2023. Accessed December 29, 2023. https://www.fda.gov/medical-devices/ medical-device-recalls/ becton-dickinson-bdcarefusion-303-recalls-alaris-infusion-pumps-due-compatibility-issues-cardinal
The sources reported no relevant financial disclosures but noted that Visante works with a broad group of companies.