Indiana University School of Medicine
Indianapolis, Indiana
This article reviews the definitions of idiopathic acute pancreatitis (IAP) and idiopathic acute recurrent pancreatitis (IRAP), etiologies to consider in initial presumed IAP and IRAP, and the role of further diagnostic modalities in determining etiology. These modalities can be noninvasive, such as magnetic resonance cholangiopancreatography (MRCP), or invasive, such as endoscopic ultrasound (EUS) and, to a lesser extent, endoscopic retrograde cholangiopancreatography (ERCP), which now has more of a therapeutic role in the treatment of selected patients with IAP and IRAP.
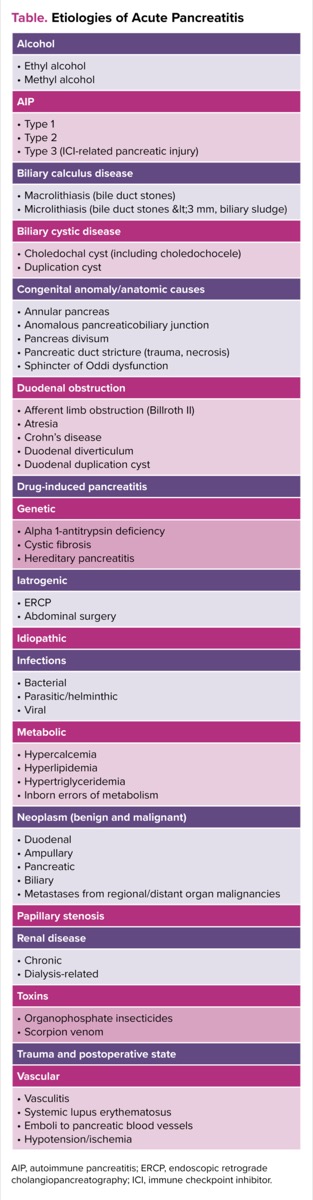
Gallstone disease and alcohol dependence cause most cases of acute pancreatitis (AP), with gallstone disease estimated to cause between 40% to 70% of cases and alcohol estimated to cause 25% to 35%.1-3 Other less common causes of AP include hypertriglyceridemia, hypercalcemia, adverse drug effects, pancreatic and ampullary neoplasia, complications of ERCP, and abdominal trauma. A comprehensive list of known causes is provided in the Table.
| Table. Etiologies of Acute Pancreatitis |
| Alcohol |
|---|
| AIP |
| Biliary calculus disease |
| Biliary cystic disease |
| Congenital anomaly/anatomic causes |
| Duodenal obstruction |
| Drug-induced pancreatitis |
| Genetic |
| Iatrogenic |
| Idiopathic |
| Infections |
| Metabolic |
| Neoplasm (benign and malignant) |
| Papillary stenosis |
| Renal disease |
| Toxins |
| Trauma and postoperative state |
| Vascular |
| AIP, autoimmune pancreatitis; ERCP, endoscopic retrograde cholangiopancreatography; ICI, immune checkpoint inhibitor. |
A significant proportion of patients with AP have no clearly identified etiology despite a thorough initial workup with laboratory tests and cross-sectional imaging. These patients, who are labeled as having presumed idiopathic acute pancreatitis (IAP), account for 10% to 30% of AP cases.4,5 A proportion of patients with IAP eventually have a confirmed etiology after a second-tier workup, which may include cross-sectional imaging with MRCP if not done during the initial evaluation, EUS, autoimmune pancreatitis evaluation, and genetic testing. Those who still do not have a clear cause elucidated at this point may then be considered to have true IAP. The proportion of patients considered to have true IAP is estimated to be closer to 10%.6 Potential factors associated with true IAP have not been conclusively elucidated, but some proposed theories include unidentified genetic polymorphisms, exposure to smoking and other environmental toxins, and effects of coexisting diseases often associated with AP, including obesity and diabetes.7
Recurrent acute pancreatitis (RAP) is defined as the occurrence of 2 or more episodes of AP with complete resolution of symptoms between episodes and without concurrent clinical or imaging evidence supportive of chronic pancreatitis (CP). When a clear etiology is not elucidated for RAP, it is referred to as idiopathic recurrent acute pancreatitis (IRAP) and can be further described as presumed or true IRAP based on the definitions above. These subcategories are distinct from smoldering pancreatitis, in which patients recovering from AP experience unremitting abdominal pain, persistently elevated pancreatic enzyme levels, with inflammatory changes in and around the pancreas seen on imaging studies, in the absence of systemic or local complications.8
Recurrence after an index episode is estimated to occur in approximately 16% to 43% of cases.9 The risk for a second or additional episodes of AP in the absence of alcohol or tobacco use is estimated to be 10% to 30%, which increases to 24% to 30% in patients with ongoing alcohol use and 40% to 50% in patients with both alcohol and tobacco use.10
In a meta-analysis by Sankaran et al, the pooled CP prevalence was 10% after 1 AP event, with a rise to 36% in patients with RAP.11 There are also data to suggest that RAP is associated with the development of exocrine and/or endocrine insufficiency.12,13 A comprehensive workup to establish an etiology is, therefore, of great importance. Determination of the etiology may alleviate patient distress over diagnostic uncertainty, aid selection of the appropriate treatment and measures to prevent recurrence, and allow estimates of the risk for complications, including progression to CP, exocrine and endocrine insufficiency, and pancreas cancer. IRAP also may lead to high healthcare resource utilization, with a financial burden and psychological distress for the patient.
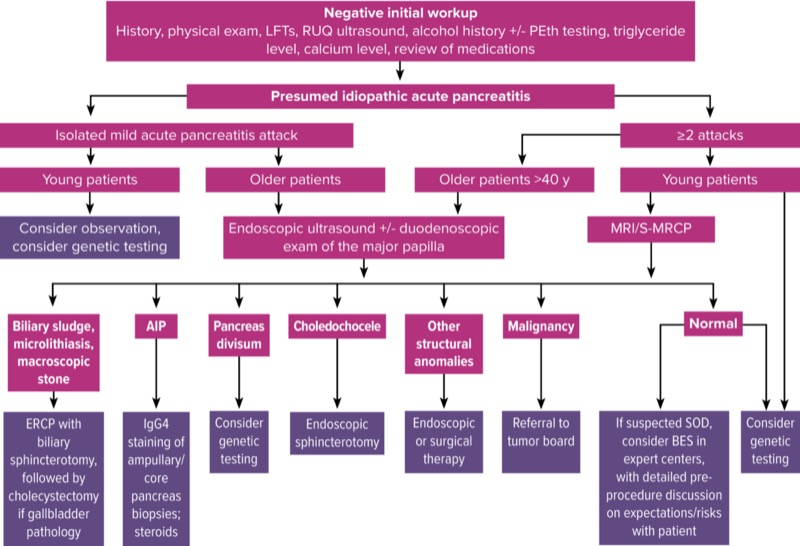
It should be emphasized, however, that the extent of evaluation should be tailored to the patient’s presentation and clearly identifiable risk factors for AP. For example, a patient who presents with 1 or more episodes of AP in the setting of persistently uncontrolled hypertriglyceridemia or ongoing alcohol use disorder may not warrant additional etiologic evaluation. Our algorithm at Indiana University for evaluation of presumed IAP and IRAP is shown in Figure 1.
Pathophysiology of AP
The majority of available data on AP pathogenesis comes from animal models. While a detailed review of proposed AP pathophysiology is outside the scope of this review, a brief summary based on work by Rinderknecht, Keck, and Bhatia, is provided.14-16 First, a triggering event, usually extrapancreatic in origin, occurs. This leads to activation of trypsinogen to trypsin in the pancreatic acinar cells. This, in turn, activates a cascade of enzymes, complement, and kinin systems. These active enzymes damage acinar cells, interstitium, and vascular endothelium. This damage to cells and tissue leads to the release of even more activated enzymes from the damaged cells and blood vessels, further amplifying and perpetuating the cycle of autodigestion. Microcirculatory injury also occurs and leads to an increase in vascular permeability and, thus, gland edema. It has been theorized that any event that causes ductal obstruction and consequent duct hypertension may result in inhibition of enzyme secretion.17 This results in colocalization of inactive pancreatic enzymes and lysosomal hydrolases with subsequent acinar cell injury.18 In cases in which there is evidence of ductal obstruction, endoscopic intervention may help relieve the obstruction, possibly preventing further episodes of pancreatitis.
Diagnostic Accuracy of MRCP and EUS
An initial workup for AP should include a comprehensive history, a thorough physical examination, laboratory testing (including liver function tests [LFTs], and measurement of calcium and triglycerides), and abdominal imaging.
Patients often underreport alcohol use, so it may be prudent to obtain objective evidence aligning with the history and additional available clues (eg, elevated mean corpuscular volume and/or aspartate aminotransferase level greater than alanine aminotransferase level). Since patients may have temporarily stopped drinking a few days before hospital presentation due to evolving abdominal pain, a blood alcohol level may not be an accurate representation of typical intake. The level of phosphatidylethanol (PEth), which can be detected in blood, hair, and urine, can be measured. The PEth blood test can detect alcohol up to 28 days after consumption.19
Commonly, patients undergo CT imaging in the emergency room at presentation, before hospital admission. However, CT may not provide specific details to aid in elucidating the etiology of the episode. A transabdominal ultrasound (TUS) may be more sensitive for identification of cholelithiasis, which might favor a biliary etiology in the appropriate setting.20,21 However, TUS may have a reduced sensitivity for detection of cholelithiasis due to ileus and bowel distension,22 and additional imaging (or repeat examination once AP has resolved) may be required. When there is intermediate probability for a biliary etiology for AP, an MRI/MRCP is done during the index admission to confirm or refute this supposition and to triage patients either to ERCP for stone extraction or conservative management.
If the above initial in-hospital workup is negative, MRI/MRCP, secretin-enhanced MRCP (S-MRCP), and EUS are the usual second-level imaging modalities deployed in the outpatient setting to continue the workup for possible etiologies. These may help to exclude occult biliary disease while providing an assessment of structural pancreatic anomalies and the presence of CP. In older adults, it is also essential to rule out periampullary or pancreatic neoplasia (further discussed below). EUS and MRCP have assumed a central role in the evaluation of patients with presumed IAP because of their high diagnostic accuracy and low morbidity.23 S-MRCP improves the diagnostic yield of MRCP for identifying underlying structural anomalies when present.24,25 When these second-level evaluation modalities were used in a multicenter study by Hallensleben et al, an etiology was detected in twice the number of IRAP patients as patients who had only had 1 IAP episode (58% vs 27%; P<0.001).26
There is no recommended timing for an MRCP or S-MRCP when there is a low probability for biliary disease during the index admission (normal LFTs, non-dilated CBD, etc). The practice at our center is to wait 4 weeks after the AP episode, which allows for resolution of pancreatic inflammation and may improve visualization. This timing also applies to EUS, as experts agree that inflammatory changes may reduce the detection of small or subtle lesions and interfere with CP assessment.27-28
Selection of imaging modality is dependent on patient age, clinical scenario, and the suspected risk factors or etiologies that are higher on the differential diagnosis of IAP. The presence of contraindications to a particular modality (eg, non–MRI-compatible cardiac pacemaker or high risk for sedation precluding EUS) may also affect this decision. A recent meta-analysis by Wan et al found that overall, EUS was superior to MRCP (64% vs 34%) for determining an eventual etiology in patients with presumed IAP.29 However, S-MRCP was superior to MRCP (without secretin) and EUS in the identification of pancreas divisum (PD).
In a study by Choudari et al presented only in abstract form, 21% of patients aged 40 to 60 years and 25% of patients older than 60 years had a neoplastic process as the cause of their AP, in contrast to only 3% of those younger than 40 years.30 On the basis of this and other studies reporting similar findings,31 the American Gastroenterological Association (AGA) recommends proceeding with EUS in the evaluation of IAP in patients aged 40 and older.1 This is our current approach as well because EUS after a first episode of IAP helps us assess for occult tumors or other structural causes of AP. With recent improvements in TUS technology, the sensitivity for detection of gallbladder stones exceeds 95%.32 However, if the TUS is negative for cholelithiasis and clinical suspicion warrants (eg, AP accompanied by elevated LFTs or preceding episodes of biliary colic), we consider EUS for this indication alone in patients of any age (see section on Occult Biliary Disease). MRCP may be considered as an alternative imaging modality in patients with contraindications to anesthesia or endoscopy, or a reluctance to undergo sedation, although it is not as sensitive as EUS for the diagnosis of occult biliary disease29 and small pancreatic lesions.33
ERCP now is used primarily as a therapeutic option, not as a diagnostic tool alone. It is best used in presumed IAP when a clear etiology has been newly identified by EUS or MRCP. Historically, ERCP was deemed to have additional diagnostic utility either for performance of sphincter of Oddi manometry (SOM) and/or aspiration of bile for crystals. These diagnostic uses are no longer routinely pursued, as discussed below.
Etiologies to Consider in IAP and IRAP
Occult Biliary Disease
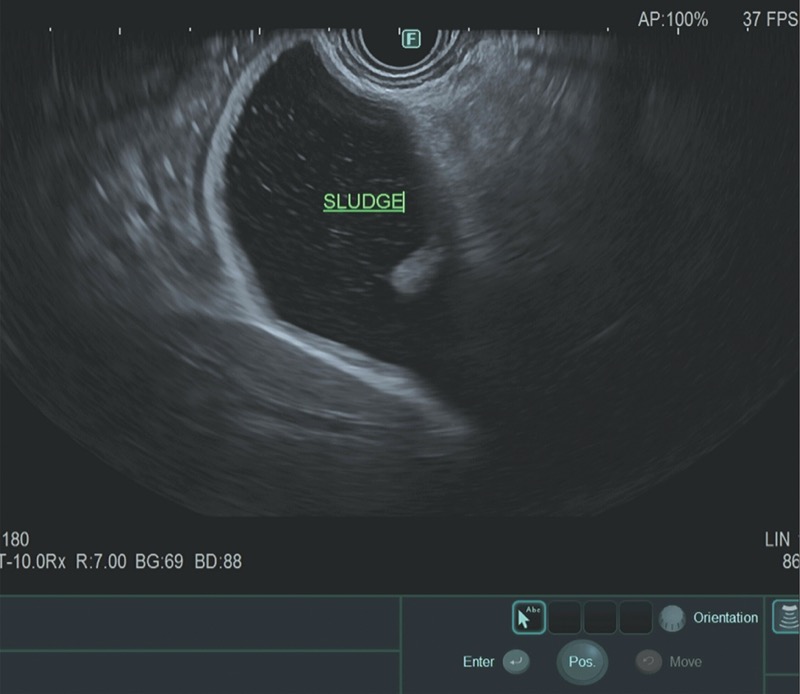
Occult biliary disease has been implicated as a common cause of IAP, with the mechanism being transient obstruction at the ampulla of Vater. Microlithiasis (stones <3 mm in diameter) and biliary sludge—a suspension of crystals (cholesterol monohydrate, calcium bilirubinate, or calcium carbonate), mucin, glycoproteins, cellular debris, and proteinaceous material within bile—can be found within the gallbladder or bile ducts and may be missed by standard imaging. Prospective studies have found that approximately one-fifth to three-fourths of patients with presumed IAP have sludge or stones in the gallbladder, 34,35 with diagnosis based on microscopic examination of bile for crystals and usually confirmed on evaluation of the resected gallbladder or follow-up gallbladder ultrasound showing gallstones and/or sludge.
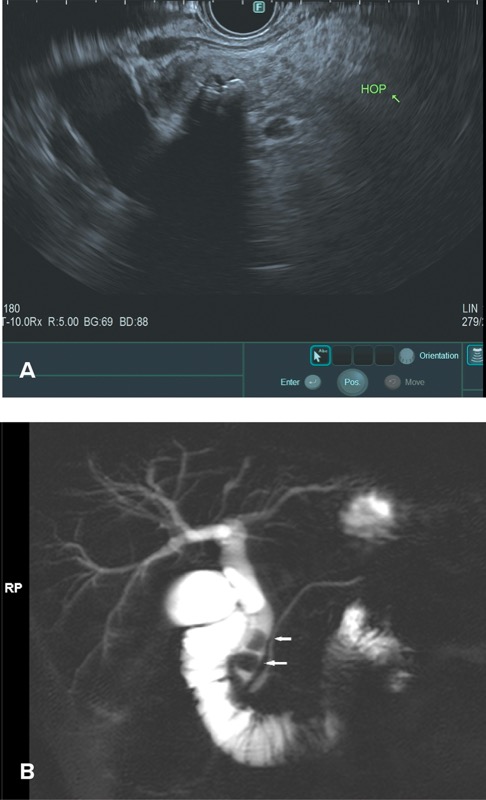
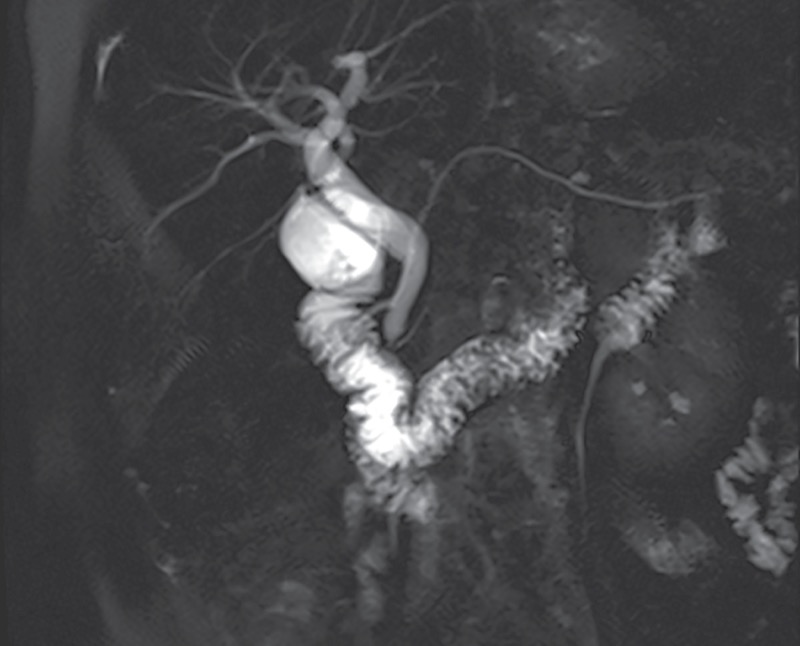
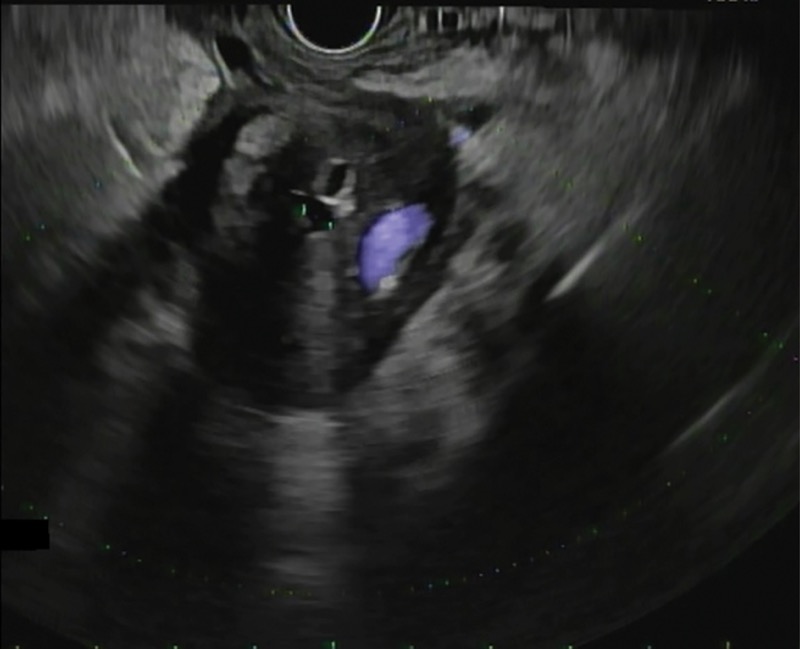
In the past, at the time of ERCP, it was recommended that bile be collected from the duodenum or bile duct after gallbladder stimulation with cholecystokinin or by direct cannulation of the gallbladder.36,37 Today, however, ERCP rarely is performed specifically for bile crystal analysis, given the risk for post-ERCP pancreatitis and the high sensitivity of EUS in detecting biliary sludge and microlithiasis (Figures 2 and 3A). Furthermore, TUS technology also has advanced significantly, with improved detection rates.
In a large meta-analysis, EUS was found to have a significant benefit in detecting occult biliary disease compared with MRCP (Figure 3B) in IAP.29 Ardengh et al found gallbladder microlithiasis in 27 of 36 patients with IAP using EUS (75%).38 When compared with the final surgical resection specimen, the sensitivity and specificity for EUS identification of gallbladder microlithiasis were 92.6% and 56%, respectively. The overall EUS accuracy in this study was 83.2%.
Intraductal ultrasonography is another modality that may be useful in detecting microlithiasis and sludge within the bile ducts, although this technique is not readily available in North America. Kim et al analyzed 31 patients with IRAP and negative findings on ERCP.39 Intraductal ultrasound revealed microlithiasis in 5 patients (16.1%) and sludge in 3 patients (9.7%).
While confirmation of a biliary etiology may have management implications (eg, proceeding with cholecystectomy), the finding of gallbladder pathology does not definitively confirm a biliary etiology of IAP. As previously noted, a patient with alcohol use disorder or marked hypertriglyceridemia who presents with AP may have a readily identifiable etiology for the episode, regardless of whether gallbladder stones or microlithiasis are identified.
A 2019 systematic review of 28 studies with 1,850 patients found only a 2% AP recurrence rate over a mean follow-up period of 20.5 months in patients with biliary disease who underwent cholecystectomy.40 Another meta-analysis of 1,679 patients by Umans et al found a 59% lower odds of recurrence of IAP (defined as episodes of AP where the etiology remained elusive despite an extensive workup including EUS or MRCP) in patients who had a cholecystectomy compared with patients managed conservatively.41 This suggests that an initially missed diagnosis of biliary pathology in the setting of AP can erroneously be labeled as IAP and that current diagnostics are insufficient to exclude a biliary cause.
When deciding if the finding of gallbladder sludge is truly the etiology of AP, it is important to keep in mind that gallbladder sludge may occur in patients who have been fasting during an AP episode. This may be the result, therefore, and not the cause of AP. Regardless, identification of microlithiasis, biliary sludge, or gallstones should prompt referral for cholecystectomy in otherwise unexplained AP, as this demonstrably reduces the odds of recurrent AP. If cholecystectomy is pursued, we recommend intraoperative cholangiography (IOC) if preoperative ERCP and biliary endoscopic sphincterotomy (BES) have not been done recently. Laparoscopic cholecystectomy’s overall favorable risk profile may influence physicians to advocate for empiric cholecystectomy, especially with repeated AP attacks. It is important, however, to have an informed discussion with the patient before a cholecystectomy about the possibility of no improvement in RAP if there are unidentified competing etiologies.
The benefit of an ERCP with empiric BES for presumed gallbladder sludge, microlithiasis or stones (ie, not clearly identified on imaging) in patients fit for surgery is controversial. There are data to suggest that patients undergoing ERCP for evaluation of AP have higher odds of developing post-ERCP pancreatitis.42 Our recommendation, therefore, is to not pursue ERCP in asymptomatic surgical candidates with a low likelihood or intermediate probability for choledocholithiasis without clearly demonstrated biliary pathology on EUS or MRCP.43 However, in patients in whom bile duct sludge or choledocholithiasis is demonstrated clearly, an ERCP with BES is recommended.
For patients with a biliary etiology who are ineligible for cholecystectomy (eg, poor surgical candidates), BES provides a therapeutic benefit to prevent recurrence. In theory, recurrent biliary pancreatitis should not occur following an adequate BES because the biliary and pancreatic orifices are separated and an impacted stone at the orifice will not lead to pancreatic duct obstruction and pancreatitis.44 Cholecystitis, cholangitis, and biliary colic, however, may still occur.
If ERCP and BES do not take place, performing cholecystectomy during the index admission diminishes the likelihood of recurrent biliary events.45
Sphincter of Oddi Dysfunction
There is still some controversy about whether a true causative effect between sphincter of Oddi dysfunction (SOD) and AP exists, given the paucity of definitive evidence to that effect. While there is evidence to suggest that there is increased sphincter tone in 30% to 65% of patients with IAP,46 it is unclear whether this sphincter hypertension leads to RAP episodes or if inflammation from the RAP episodes is what leads to sphincter hypertension.
Previously, some physicians believed that pancreatic SOD might be responsible for many cases of RAP in which a thorough etiologic evaluation was negative. Historically, endoscopic sphincterotomy via ERCP was done to reduce the basal pressure of the sphincter of Oddi. Biliary endoscopic sphincterotomy was the initial approach taken, based on limited data.47 Subsequently, Guelrud et al demonstrated a reduced frequency of RAP episodes in a small cohort after pancreatic endoscopic sphincterotomy.48 This finding was supported by similar results in three other small cohort studies,36,49,50 so practice subsequently shifted to a combined pancreaticobiliary or dual endoscopic sphincterotomy.
Due to the lack of high-level evidence of benefit from this new approach, Cote et al conducted a randomized clinical trial to evaluate ERCP with SOM and sphincterotomy in patients with IRAP with or without an increase in basal pancreatic sphincter pressure.46 Patients were followed for a median of 78 months. In 69 patients with pancreatic SOD, the frequency of RAP during the follow-up evaluation was similar among those randomized to biliary or dual sphincterotomy (48.5% vs 47.2%; P=1.0).
This study generated further controversy among clinicians, with some concluding that ERCP and sphincterotomy should not be offered to or performed in patients with IRAP because half the patients studied continued to present with RAP post-therapy. However, proponents of ERCP noted that half the patients studied did not present with further RAP episodes, with nearly 7-year follow-up. Furthermore, a subsequent post hoc analysis of these data demonstrated a reduction in RAP episode density in those who presented with recurrent episodes during the long follow-up period.51
BES alone may be considered in patients with IRAP in whom SOD is strongly suspected. It is important, however, to have a thorough discussion with patients on risk versus benefit, as well as to set patients’ expectations before proceeding with endoscopic therapy for several reasons. First, while a post hoc analysis of longer-term data from the above study by Cote et al,46 orally presented in abstract form,51 suggests that there may be long-term benefit in reducing the frequency of AP attacks, a more recent study by the same author shows there is still a relatively high recurrence rate of AP (regardless of the type of endoscopic sphincterotomy performed) in the short term.52 Furthermore, this patient population has also been demonstrated to be at higher risk for post-ERCP pancreatitis.42 More high-quality randomized trials are needed to definitively determine if there is still a role for pancreatic sphincter therapy in patients with a normal pancreatogram at ERCP, particularly given the high restenosis rates observed.53
Pancreas Divisum
PD, the failure of the dorsal and ventral buds to fuse during organogenesis, is the most frequent congenital variation of pancreatic anatomy. It results in drainage of most of the pancreatic exocrine juice through the dorsal pancreatic duct and into the duodenum through the minor papilla.54 Autopsy series suggest that PD is found in 5% to 10% of the general population.55-58
It remains controversial, however, whether PD is a primary cause, a predisposing factor, or an innocent bystander in the development of RAP, especially as studies suggest that only 5% of subjects with PD present with symptomatic pancreas disease. Conversely, up to 19% of patients with presumed IRAP have been found to have PD.31
There is also a demonstrated increased prevalence of genetic abnormalities in patients with PD and RAP,44,59-62 with cystic fibrosis transmembrane conductance regulator (CFTR) gene mutations identified in approximately 47% of patients with PD and RAP.44,61 This suggests a synergistic effect of structural anomalies and genetics to induce AP. For patients with PD who have symptomatic pancreas disease, a potential mechanism may be pancreatic duct hypertension due to obstruction of pancreatic exocrine juice flow through the minor papilla, which in turn precipitates RAP. However, this presumably does not apply in all patients because the majority of patients with RAP and PD are found to have a non-dilated dorsal pancreatic duct when evaluated by MRCP or ERCP.63 CFTR mutations likely increase pancreatic juice viscosity and acidity via impaired ion transport, exacerbating the impaired outflow. There is an excessive intrapancreatic inflammatory response, reduction in apoptosis, and disruption of intracellular signaling and mitochondrial function. These exacerbate pancreatic injury during AP episodes.64
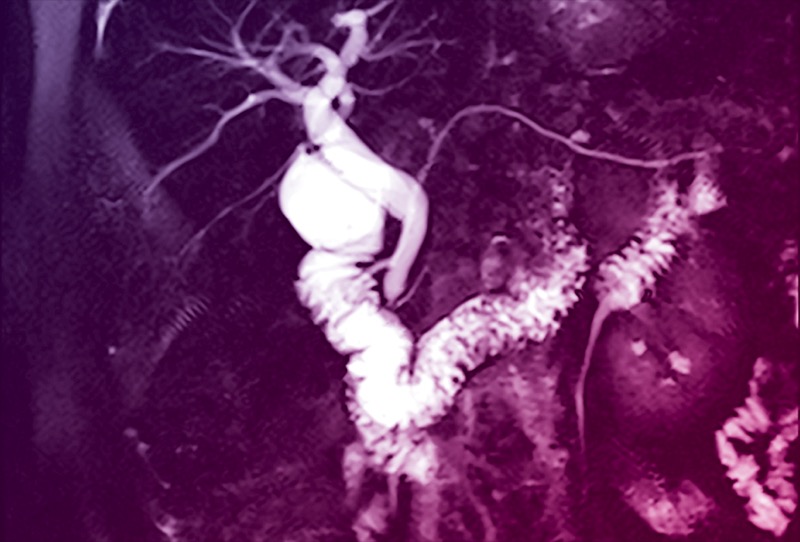
MRCP and S-MRCP are usually the diagnostic modalities of choice for PD, given their noninvasiveness (Figure 4). Studies have demonstrated the high diagnostic accuracy of S-MRCP.65-67 Mosler et al found that S-MRCP had an overall sensitivity and specificity of 73% and 97%, respectively, when compared with ERCP in 19 patients.65 The sensitivity and specificity improved to 83% and 99% in the subgroup of patients without CP. A meta-analysis and systematic review of 11 studies evaluating the utility of MRCP, S-MRCP, and EUS found similar results, with specificities of 99% for both MRCP and S-MRCP but a higher sensitivity of S-MRCP (83% vs 59%).66 Data also suggest that S-MRCP could help identify a subgroup of patients who might respond to endoscopic therapy, as evidenced by a recently published retrospective study from 3 tertiary centers in China evaluating 120 patients with PD who underwent endoscopic retrograde pancreatography.67 The investigators found that linear array EUS had a sensitivity of 90.8%, a specificity of 99.8%, and an overall diagnostic accuracy of 99%, suggesting it is a reasonable alternative to S-MRCP. They also noted that the degree of duct dilation suggested an obstructive process and might correlate with or predict a response to minor papilla intervention, but this assumption has not been proven conclusively to date.
From a management standpoint, there has been a lack of high-quality evidence regarding the efficacy of endoscopic therapy. Small cohort studies have evaluated the effect of various combinations of therapies, including minor papilla endoscopic sphincterotomy (MiES), minor papilla dilation, repeated dorsal duct stenting, or a combination of these.68-71 While a review of these trials suggests a potential benefit from endoscopic therapy, with 68% to 90% of patients having no further episodes of AP after intervention, it needs to be emphasized that the mean follow-up interval was 30 months.44 Clearly, this is insufficient in an episodic illness (ie, RAP), when attacks may be separated by several years.
A meta-analysis of 323 PD patients from 13 retrospective studies treated with MiES showed an overall success rate of 77%, defined as no further episodes of RAP, reduction of episodes of RAP, or improvement in quality of life.72 When the subgroup of studies in this meta-analysis with a clear clinical success definition (“no further AP in the follow-up”) were evaluated, the pooled proportion achieving clinical success was 69.8%. When studies with other definitions of clinical success (reduction of RAP episodes, improvement in quality of life) were evaluated, the clinical success rate was 81.2%.
In the only randomized controlled trial published to date evaluating dorsal pancreatic duct stent placement for RAP in patients with PD, Lans et al demonstrated a remarkable improvement in symptoms for 90% of treated patients versus 11% of controls, with a follow-up period of 12 months.68
However, systematic reviews and meta-analyses suggest a lower response rate for endotherapy in PD in the range of 62% to 70%.69,73-76 In these studies, patients with RAP showed the most improvement in symptoms (75%-80%), followed by patients with CP (42%-69%). Patients with CP-like abdominal pain showed the least improvement (33%-54%). There were notable adverse events, including a post-ERCP pancreatitis incidence of 10% to 20% and a 19% incidence of minor papilla stenosis requiring re-intervention.74-76 The mean follow-up period for these studies was 32 months, with most having a 36-month follow-up window.54,69,73-76
This is problematic because patients often can remain asymptomatic for several years between pain episodes or pancreatitis flares but could potentially require reintervention at a much later time.77 Also, dorsal duct stent placement requires repeated procedures for stent change, each with an associated risk for post-ERCP pancreatitis, as well as the ductal and parenchymal changes that may occur after pancreatic duct stent placement and may be irreversible.78,79
Advocates of endoscopic therapy, therefore, have favored performance of MiES as a better option to enlarge the minor papilla orifice.44,80 The short-term follow-up of these studies was the impetus for the SHARP trial, a randomized sham controlled trial evaluating the efficacy of MiES in patients with PD and RAP.81 This study was recently completed, with a reported median follow-up period of 33.5 months. Reported pre-publication results suggest that minor papilla endotherapy does not lead to a significant reduction in recurrent episodes of AP when compared with sham.82 This may lead to a practice change in the near future, with most patients no longer being offered endoscopic therapy.
Choledochal Cysts
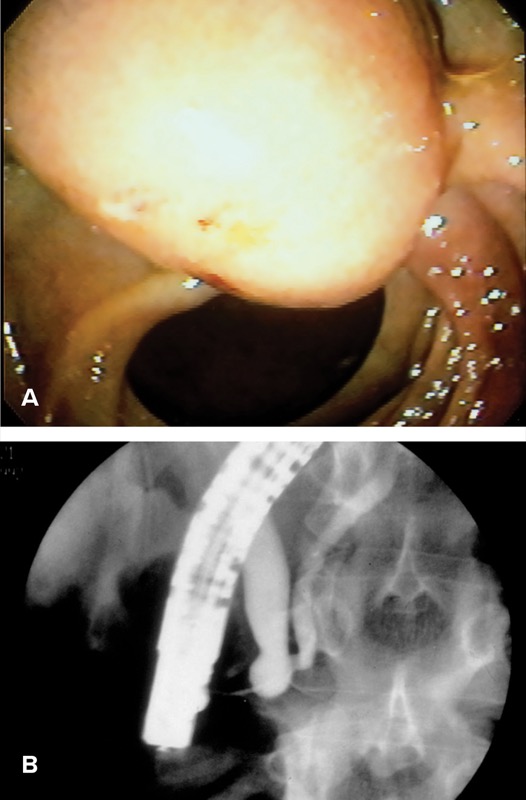
Congenital pancreaticobiliary malformations, often incidentally found on MRI/MRCP, can lead to RAP. Choledochal cysts are cystic dilations of the intrahepatic or extrahepatic biliary tree and may be single or multiple. Todani type III choledochal cysts, or choledochoceles, are cystic lesions that arise from the intramural bile duct or intra-ampullary common channel (Figure 5A, 5B).83 In type A choledochoceles, as originally defined by Sarris and Tsang,84 the intramural bile duct communicates with a cystically dilated segment, which then drains into the duodenal lumen via the separate papillary orifice.85,86 In type B choledochoceles, the bile duct drains normally into the duodenum, with the choledochocele arising as a diverticulum from the intra-ampullary common channel.44,87
During ERCP, the papilla associated with the type A choledochocele may be recognized by its bulging intramural portion of the bile duct, but it is soft when probed with a catheter tip.88 Alternatively, the papilla may appear less prominent but progressively enlarges with contrast injection that fills the cyst. Type B choledochoceles often appear endoscopically as a pendulous duodenal mass, with the ampullary orifice immediately proximal to this mass.87
Choledochoceles commonly present with pancreatitis (38%-70%),44,89 but they are an uncommon overall cause of IAP due to their low prevalence.89 The most widely accepted mechanism of AP in patients with choledochoceles is obstruction of outflow with subsequent ductal hypertension, or bile reflux into the pancreatic duct.90
MRCP can detect choledochoceles with reasonable accuracy. In a study of 72 patients using ERCP as the gold standard, the sensitivity and specificity of MRCP in diagnosing type III choledochal cysts was 73% and 100%, respectively.91
EUS is also a viable diagnostic modality, but its utility in the diagnosis of choledochoceles is limited to case reports.92,93
While surgical therapy (either excision or sphincteroplasty) has been the historical approach to choledochoceles, endoscopic therapy with ERCP is considered to be a safe and effective alternative in most patients.87 Endoscopic management involves either unroofing the cyst, BES, snare resection, or a combination of the above, depending on cyst type and endoscopist preference. A review of published case reports suggests a resolution of AP episodes in 13 of 14 cases (93%) after endoscopic intervention.85,94-97 Anomalous pancreaticobiliary junction is rare, occurring in 0.2% of European populations, but is more prevalent in Asian populations and often is associated with choledochal cysts (40%-70%).44,98 Patients with anomalous pancreaticobiliary junction may experience AP through bile reflux into the pancreatic duct, and BES can be beneficial in preventing further AP episodes.
Although choledochal cysts are associated with increased risk for biliary tract malignancy, malignancy complicating a choledochocele rarely is seen and has been limited to case reports.44 Malignancy can develop after endoscopic therapy, but surveillance protocols have not been defined.
Other Anatomic Causes
A number of non-neoplastic structural lesions of the pancreas have been associated with AP. These can be evaluated with ERCP, EUS, and/or MRCP and are listed in the Table.
Five to seven percent of patients with benign or malignant pancreatobiliary and ampullary tumors present with IAP.99 These lesions can be solid or cystic and should be considered in the differential diagnosis of patients 40 years or older presenting with IAP. The presumed pathophysiology for these conditions, whether benign or malignant, is obstruction of the pancreatic duct.
Mucinous Tumors
Mucinous tumors of the pancreas, both intraductal papillary mucinous neoplasms (IPMNs) and mucinous cystic neoplasms (MCNs), may present as RAP.31,100-102 Unlike IPMNs, MCNs rarely communicate with the pancreatic duct, and, thus, the presenting symptoms of MCNs are often due to compression of adjacent structures or organs. Rarely, MCNs can precipitate AP by extrinsic compression of the pancreatic duct. On the other hand, both side-branch and main-duct IPMNs may lead to AP through mucin production and, thus, duct obstruction. Both mucinous lesions are premalignant, with varying malignant potential, typically based on high-risk imaging findings such as mural nodules or associated solid components, diffuse or focal dilation of the main pancreatic duct greater than 5 mm, size of 3 cm or more, or continued increase in cyst size of 3 mm or more per year.103,104
Mucinous cysts typically are surveilled with MRI/MRCP, EUS, or both in an alternating fashion, based on the presence or absence of high-risk features and, if available, cyst fluid analysis. ERCP has no role in the diagnosis of MCNs but on occasion may be helpful in IPMN diagnosis and management planning. Rarely, an IPMN may be identified only by subtle intraductal cast-like filling defects (eg, mucin) seen with good ERCP technique, a finding that MRCP could have missed, particularly if S-MRCP had not been performed. Indeed, it is not uncommon to find patients with an IPMN presenting with RAP for many years before the diagnosis has been made.105 Similarly, it may be difficult to determine whether a patient who presents after several RAP episodes and is found to have a small pancreatic cyst and a prominent but non-dilated pancreatic duct has an IPMN or CP.
EUS-guided cyst or pancreatic duct aspiration has been associated with postoperative pancreatitis, with one study finding an up to 8% incidence of post EUS-FNA pancreatitis in patients with branch-duct IPMNs compared with 1.3% with other cyst types.106 ERCP may be considered as an alternative in such cases, particularly for main-duct IPMNs, perhaps with pancreatoscopy for more detailed analysis. This may be dependent on local expertise.
When an IPMN results in RAP, this is considered a worrisome feature that warrants further evaluation with consideration of cyst fluid sampling, as well as multidisciplinary discussion regarding the need for possible surgical resection, per the 2018 American College of Gastroenterology guidelines.103
Solid Tumors
Solid tumors (benign and malignant) are identified as the etiology in 5% to 7% of AP cases initially believed to be idiopathic99 and should be suspected in older patients as well as those with preexisting constitutional symptoms or new-onset diabetes accompanying AP. A cohort study using a representative database from the United States found that 5.9% of patients with an eventual diagnosis of pancreatic cancer in their cohort had a sentinel event of AP within 90 days of diagnosis.107
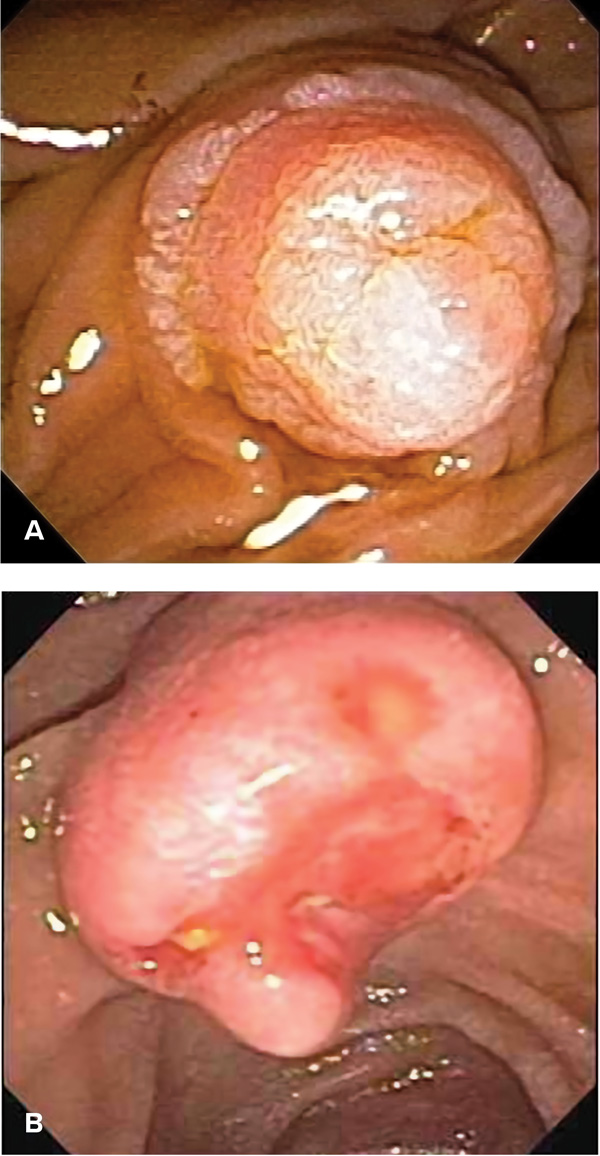
Ampullary tumors, either benign (Figure 6A) or malignant (Figure 6B), also may present with AP. A wide variety of benign tumors can arise at the major papilla, including adenoma, lipoma, fibroma, lymphangioma, leiomyoma, and hamartoma, with adenomas being the most common.108 Patients with hereditary conditions such as familial adenomatous polyposis may have duodenal adenomas involving the major papilla, which can lead to presentation at a young age with AP.
Endoscopic therapy is the treatment of choice for most ampullary adenomas (snare papillectomy, thermal ablation, or a combination of the two).109-111 Primary malignant tumors of the major papilla include carcinoma, lymphoma, and neuroendocrine tumors.110 Although most patients with malignant tumors of the papilla present with obstructive jaundice, occasionally patients develop AP as the first sign of their disease.
Lastly, metastases to the pancreas from other organs can occur, with renal cell carcinoma, lung cancer, breast cancer, and melanoma most frequently seen, although AP due to pancreatic duct obstruction is an uncommon complication. Uroepithelial malignancies, sarcomas, and lymphoma also may metastasize to the pancreas112-114 and potentially cause AP due to duct obstruction.
While CT and MRI/MRCP have clear utility in this scenario, EUS outperforms cross-sectional imaging from a diagnostic standpoint, as it has a higher sensitivity for smaller (1-3 cm) lesions33 and has the added advantage of same-session tissue acquisition for a definitive diagnosis. EUS, however, does not provide additional staging information above that provided by a good-quality, contrast-enhanced, thin-cut CT scan.115 As noted earlier, given the increased prevalence of neoplasia in older adults with IAP and the importance of an early diagnosis, the AGA recommends EUS for evaluation in patients 40 years and older, even in the absence of red flags.1 The primary role of ERCP in these patients is therapeutic because biliary stent placement in malignant biliary obstruction can be offered to unresectable patients or surgical candidates receiving neoadjuvant chemotherapy.116 With ampullary lesions, targeted biopsies are best obtained with side-viewing endoscopy.
Genetic Mutations
The prevalence of genetic mutations in IRAP is thought to be approximately 30% to 60%.117,118 The true incidence of genetic mutations, however, is almost certainly underestimated because routine genetic evaluation after an episode of IAP has yet not been adopted into widespread clinical practice. Mutations of the cationic trypsinogen gene (serine protease 1 gene; PRSS1) are associated with hereditary or familial pancreatitis,119 onset of AP in childhood, and frequent progression to CP.120,121 Serine protease inhibitor Kazal type 1 (SPINK1) mutations have been detected in 16% to 23% of patients with apparent IAP, compared with only about 2% of healthy controls,122 supporting an association. CFTR gene mutations are associated with the most common inherited disease of the exocrine pancreas. Although CFTR gene mutations occur in 5% of White European and North American populations, IAP may occur in up to 20% of CFTR heterozygotes.123 Chymotrypsin C (CTRC) variants have been shown to increase the risk for rapid progression to CP.124 There are other genetic mutations that have been associated with IAP, but a full assessment of the role of genetic polymorphisms is outside of the scope of this review.
A 2015 study by Culetto et al prospectively investigated 309 IAP patients and identified genetic mutations in 10% of patients 35 years of age or younger, compared with 1.5% of patients older than age 35.125 Another study found that mutations in 3 genes (CFTR, SPINK1 and PRSS1) were present in up to 32% of young patients (<30 years of age) with IAP.126 In another retrospective single-center study evaluating 97 IRAP patients, all of whom underwent genetic testing,118 56 patients (58%) had 1 or more pathogenic variants, with CFTR being the most common (48), followed by SPINK1 in 22, PRSS1 in 5, and CTRC in 2 patients. Among patients in that cohort who were younger than age 35 and had 1 unexplained episode of AP, 63% had at least 1 pathogenic variant identified. Multiple logistic regression analysis identified both IRAP (odds ratio [OR], 18.2; P=0.008) and IAP (OR, 2.46; P=0.017) in patients younger than age 35 were independently associated with pathogenic variants. Based on the above evidence, it is reasonable to recommend genetic testing for patients with IAP, particularly if they are less than age 35.
Genetic testing can be performed on blood or saliva. There are few available options for intervention in genetic-associated IAP, although CFTR gene modulator medications appear to have some utility in the management of CF heterozygote-related RAP.127 Identification of pathogenic mutations may help establish a cause-and-effect relationship, allowing a more informed prognostic assessment of the risk for future RAP, subsequent progression to CP, and additional complications of the disease, including pancreatic adenocarcinoma. Genetic counseling is important for both patients and family members. While ERCP and pancreatic endotherapy may still be considered in patients with a genetic etiology, it should be anticipated that this therapy will be unlikely to result in long-lasting or durable benefit. Supportive measures, including medications for symptom control, should be continued, and timely surgical evaluation, including candidacy for total pancreatectomy with islet auto transplantation in selected candidates, may be considered to reduce or eliminate the future risk for RAP and malignant transformation.128
Autoimmune Pancreatitis
Autoimmune pancreatitis (AIP) is an uncommon cause of IAP and IRAP.129 It is a chronic inflammatory disease of the pancreas (Figure 7). Historically, 2 main types of AIP have been described, with a third type associated with the use of immune checkpoint inhibitors recognized more recently. Various diagnostic criteria from different countries have been proposed to make the diagnosis of AIP.130-131 An international consensus on diagnosis initially subdivided AIP into type 1 and type 2.132,133
Type 1 AIP often is characterized by diffuse enlargement of the pancreas on imaging, with segmental irregular narrowing of the main pancreatic duct, typically without upstream pancreatic duct dilation. MRI/MRCP has a sensitivity of 84% and a specificity of 97% (compared with a sensitivity of 59% and specificity of 99% for CT) in the diagnosis of AIP.134 EUS has a higher sensitivity of 93% and a specificity of 99.3%, particularly in cases when an irregular, narrow main pancreatic duct in association with duct wall thickening is seen.135 Type 1 AIP is associated with the presence of several other autoantibodies and elevated levels of serum immunoglobulin G-4 (IgG4) subtype on serology. Fibrotic changes with lymphoplasmacytic infiltration are seen histologically.129 An international multicenter survey showed that obstructive jaundice was a more frequent presentation in type 1 versus type 2 (75% vs 47%; P<0.001), whereas abdominal pain (41% vs 68%; P<0.001) and AP (5% vs 34%; P<0.001) were more frequent in type 2.136
Type 2 AIP usually presents without multiorgan involvement and typically occurs in younger patients. There is no gender predilection, and it is associated with inflammatory bowel disease in up to 30% of cases.137 It does not have the classic elevation of serum IgG4, and, thus, a negative serologic test is not sufficient to exclude a diagnosis of AIP when there is significant clinical suspicion. The inflammatory infiltrate in type 2 AIP is typically periductal, with identification of granulocytic epithelial lesions rather than the IgG4-positive plasma cells seen in type 1 AIP.
Type 1 and type 2 AIP respond dramatically to steroids. Relapse after steroid therapy is frequent in type 1 AIP, often requiring repeat steroid therapy or the addition of steroid-sparing agents or other immunomodulators, such as rituximab. Relapse is uncommon in type 2 AIP. Laboratory screening with serum immunoglobulins with IgG subtypes and, if necessary, core biopsies of the pancreas obtained at EUS should be considered in selected IAP patients with clinical or radiological features suggestive of AIP.138 Positive IgG4 immunostaining of major papilla biopsies via the use of a side-viewing duodenoscope are specific for AIP, with a reported range of 89% to 100%, but this finding has a lower sensitivity (52%-80%).139,140 Such immunostaining is, therefore, a reasonable alternative when there is a strong suspicion for AIP but serum IgG4 levels are normal and pancreatic tissue samples cannot be obtained.
Conclusion
IAP and IRAP represent a challenging clinical problem. EUS and MRCP have assumed a central role in the evaluation of patients with these conditions, and an etiology can be found in a significant proportion of patients using second-line imaging modalities, laboratory testing (including genetic evaluation), and/or histologic evaluation of pancreas tissue. From an imaging standpoint, EUS has assumed a central role in the evaluation of IAP and IRAP, particularly with patients older than 40 years of age, with S-MRCP being an invaluable imaging modality. As a result, the role of ERCP as a diagnostic modality has greatly diminished, but it has retained a defined but important role as a therapeutic option in selected cases. Patients with IAP and IRAP are best evaluated in centers where specialized pancreatic expertise and advanced endoscopic methods are available. The algorithm we recommend in our unit is shown in Figure 1.
Research gaps remain in this challenging clinical scenario, and future high-quality multicenter studies evaluating optimal diagnostic modalities and interventions will help determine the optimal cost-effective approach for these patients.
References
- Forsmark CE, Baillie J, AGA Institute Clinical Practice and Economics Committee, et al. AGA Institute technical review on acute pancreatitis. Gastroenterology. 2007;132(5):2022-2044.
- Peery AF, Crockett SD, Murphy CC, et al. Burden and cost of gastrointestinal, liver, and pancreatic diseases in the United States: update 2018. Gastroenterology. 2019;156(1):254-272.
- Boxhoorn L, Voermans RP, Bouwense SA, et al. Acute pancreatitis. Lancet. 2020;396(10252):726-734.
- Chen Y, Zak Y, Hernandez-Boussard T, et al. The epidemiology of idiopathic acute pancreatitis, analysis of the nationwide inpatient sample from 1998 to 2007. Pancreas. 2013;42(1):1-5.
- Zilio MB, Eyff TF, Azeredo-Da-Silva ALF, et al. A systematic review and meta-analysis of the aetiology of acute pancreatitis. HPB (Oxford). 2019; 21(3):259-267.
- Somani P, Navaneethan U. Role of ERCP in patients with idiopathic recurrent acute pancreatitis. Curr Treat Options Gastroenterol. 2016;14(3):327-339.
- Forsmark CE, Vege SS, Wilcox CM. Acute pancreatitis. N Engl J Med. 2016;375(20):1972-1981.
- Testoni PA. Endoscopic pancreatic duct stent placement for inflammatory pancreatic diseases. World J Gastroenterol. 2007;13(45):5971-5978.
- Aronen A, Guilabert L, Hadi A, et al. Idiopathic acute pancreatitis (IAP)-a review of the literature and algorithm proposed for the diagnostic work-up of IAP. Transl Gastroenterol Hepatol. 2024;9:71.
- Guda NM, Trikudanathan G, Freeman ML. Idiopathic recurrent acute pancreatitis. Lancet Gastroenterol Hepatol. 2018;3(10):720-728.
- Sankaran SJ, Xiao AY, Wu LM, et al. Frequency of progression from acute to chronic pancreatitis and risk factors: a meta-analysis. Gastroenterology. 2015;149(6):1490-1500.
- Hollemans RA, Hallensleben NDL, Mager DJ, et al. Pancreatic exocrine insufficiency following acute pancreatitis: systematic review and study level meta-analysis. Pancreatology. 2018;18(3):253-262.
- Hart PA, Bradley D, Conwell DL, et al. Diabetes following acute pancreatitis. Lancet Gastroenterol Hepatol. 2021;6(8):668-675.
- Rinderknecht H. Activation of pancreatic zymogens: normal activation, premature intrapancreatic activation, protective mechanisms against inappropriate activation. Dig Dis Sci. 1986;31:314-321.
- Keck T, Friebe V, Warshaw AL, et al. Pancreatic proteases in serum induce leukocyte-endothelial adhesion and pancreatic microcirculatory failure. Pancreatology 2005;5:241–250.
- Bhatia M, Brady M, Shokuhi S, et al. Inflammatory mediators in acute pancreatitis. J Pathol. 2000;190:117-125.
- Siqin D, Wang C, Zhou Z, Li Y. The key event of acute pancreatitis: pancreatic duct obstruction and bile reflux, not a single one can be omitted. Med Hypotheses. 2009 May;72(5):589-91. doi: 10.1016/j.mehy.2008.12.009. Epub 2009 Jan 14. PMID: 19147295.
- Cosen-Binker LI, Gaisano HY. Recent insights into the cellular mechanisms of acute pancreatitis. Can J Gastroenterol. 2007;21(1):19-24.
- Fipps DC, Meyer R, Woods J, et al. Clinical utility and impact of phosphatidylethanol testing in liver transplantation evaluations. J Acad Consult Liaison Psychiatry. 2024;65(2):157-166.
- Tenner S, Dubner H, Steinberg W. Predicting gallstone pancreatitis with laboratory parameters:a meta-analysis. Am J Gastroenterol. 1994;89(10):1863-1866.
- Fogel EL, Sherman S. ERCP for gallstone pancreatitis. N Engl J Med. 2014;370(2):150-157.
- Surlin V, Saftoiu A, Dumitrescu D. Imaging tests for accurate diagnosis of acute biliary pancreatitis. World J Gastroenterol. 2014;20(44):16544-16549.
- Al-Haddad M, Wallace MB. Diagnostic approach to patients with acute idiopathic and recurrent pancreatitis, what should be done? World J Gastroenterol. 2008;14(7):1007-1010.
- Mariani A, Arcidiacono PG, Curioni S, et al. Diagnostic yield of ERCP and secretin-enhanced MRCP and EUS in patients with acute recurrent pancreatitis of unknown aetiology. Dig Liver Dis. 2009;41(10):753-758.
- Sherman S, Freeman ML, Tarnasky PR, et al. Administration of secretin (RG1068) increases the sensitivity of detection of duct abnormalities by magnetic resonance cholangiopancreatography in patients with pancreatitis. Gastroenterology. 2014;147(3):646-654 United European Gastroenterol J. 2020;8(3):340-350.
- Hallensleben ND, Umans DS, Bouwense SA, et al. The diagnostic work-up and outcomes of ‘presumed’ idiopathic acute pancreatitis: a post-hoc analysis of a multicentre observational cohort. United European Gastroenterol J. 2020;8(3):340-350.
- Rana SS, Bhasin DK, Rao C, et al. Role of endoscopic ultrasound in idiopathic acute pancreatitis with negative ultrasound, computed tomography, and magnetic resonance cholangiopancreatography. Ann Gastroenterol. 2012;25(2):133-137.
- Wilcox CM, Seay T, Kim H, et al. Prospective endoscopic ultrasound-based approach to the evaluation of idiopathic pancreatitis: causes, response to therapy, and long-term outcome. Am J Gastroenterol. 2016;111(9):1339-1348.
- Wan J, Ouyang Y, Yu C, et al. Comparison of EUS with MRCP in idiopathic acute pancreatitis: a systematic review and meta-analysis. Gastrointest Endosc. 2018;87(5):1180-1188.
- Choudari CP, Fogel EL, Sherman S, et al. Idiopathic pancreatitis: yield of ERCP correlated with patient age. Am J Gastroenterol. 1998;93:1654. Abstract.
- Fischer M, Hassan A, Sipe BW, et al. Endoscopic retrograde cholangiopancreatography and manometry findings in 1,241 idiopathic pancreatitis patients. Pancreatology. 2010;10(4):444-452.
- Wu X, Li K, Kou S, et al. The accuracy of point-of-care ultrasound in the detection of gallbladder disease: a meta-analysis. Acad Radiol. 2024;31(4):1336-1343.
- Kitano M, Yoshida T, Itonaga M, et al. Impact of endoscopic ultrasonography on diagnosis of pancreatic cancer. J Gastroenterol. 2019;54(1):19-32.
- Ros E, Navarro S, Bru C, et al. Occult microlithiasis in ‘idiopathic’ acute pancreatitis: prevention of relapses by cholecystectomy or ursodeoxycholic acid therapy. Gastroenterology. 1991;101(6):1701-1709.
- Lee SP, Nicholls JF, Park HZ. Biliary sludge as a cause of acute pancreatitis. N Engl J Med. 1992;326(9):589-593.
- Kaw M, Brodmerkel GJ Jr. ERCP, biliary crystal analysis, and sphincter of Oddi manometry in idiopathic recurrent pancreatitis. Gastrointest Endosc. 2002;55(2):157-162.
- Rashdan A, Fogel E, McHenry L Jr, et al. Frequency of biliary crystals in patients with suspected sphincter of Oddi dysfunction. Gastrointest Endosc. 2003;58(6):875-878.
- Ardengh JC, Malheiros CA, Rahal F, et al. Microlithiasis of the gallbladder: role of endoscopic ultrasonography in patients with idiopathic acute pancreatitis. Rev Assoc Med Bras. 2010;56(1):27-31.
- Kim HS, Moon JH, Choi HJ, et al. The role of intraductal US in the management of idiopathic recurrent pancreatitis without a definite cause on ERCP. Gastrointest Endosc. 2011;73(6):1148-1154.
- Pereira R, Eslick G, Cox M. Endoscopic ultrasound for routine assessment in idiopathic acute pancreatitis. J Gastrointest Surg. 2019;23(8):1694-1700.
- Umans DS, Hallensleben ND, Verdonk RC, et al. Recurrence of idiopathic acute pancreatitis after cholecystectomy: systematic review and meta-analysis. Br J Surg. 2020;107(3):191-199.
- Beran A, Aboursheid T, Ali AH, et al. Predictors of post-endoscopic retrograde cholangiopancreatography pancreatitis: a comprehensive systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2024;Dec16:S1542-3565(24)01097-8.
- ASGE Standards of Practice Committee, Buxbaum JL, Abbas Fehmi SM, et al. ASGE guideline on the role of endoscopy in the evaluation and management of choledocholithiasis. Gastrointest Endosc. 2019;89(6):1075-1105.
- Yadlapati S, Gutta A, Fogel EL. Determining the value of endoscopic retrograde cholangiopancreatography in the management of patients with acute pancreatitis and related complications. Expert Rev Gastroenterol Hepatol. Published online February 17, 2025. doi:10.1080/17474124.2025.2464057
- Moody N, Adiamah A, Yanni F, et al. Meta-analysis of randomized clinical trials of early versus delayed cholecystectomy for mild gallstone pancreatitis. Br J Surg. 2019;106(11):1442-1451.
- Cote GA, Imperiale TF, Schmidt SE, et al. Similar efficacies of biliary, with or without pancreatic, sphincterotomy in treatment of idiopathic recurrent acute pancreatitis. Gastroenterology. 2012;143(6):1502-1509.
- Lans JL, Parikh NP, Geenen JE. Application of sphincter of Oddi manometry in routine clinical investigations. Endoscopy. 1991;23(3):139-143.
- Guelrud M, Plaz J, Mendoza S, et al. Endoscopic treatment in type II pancreatic sphincter dysfunction. Gastrointest Endosc. 1995;41(4):398. Abstract.
- Toouli J, Di Francesco V, Saccone G, et al. Division of the sphincter of Oddi for treatment of dysfunction associated with recurrent pancreatitis. Br J Surg. 1996;83(9):1205-1210.
- Okolo PI, 3rd, Pasricha PJ, Kalloo AN. What are the long-term results of endoscopic pancreatic sphincterotomy? Gastrointest Endosc. 2000;52(1):15-19.
- Easler JS, Sherman S, Lehman GA, et al. Change in Incidence RATES of idiopathic recurrent acute pancreatitis following ERCP with sphincter of Oddi manometry: post-hoc analysis with long term follow-up of a randomized, clinical trial. Gastrointest Endosc. 2015 ;81(5 suppl):AB160-AB161.
- Cote GA, Elmunzer BJ, Nitchie H, et al. Sphincterotomy for biliary sphincter of Oddi disorder and idiopathic acute recurrent pancreatitis: the RESPOnD longitudinal cohort. Gut. 2024;74(1):58-66.
- Dufault DL, Elmunzer BJ, Cotton PB, et al. Adverse events and reinterventions following pancreatic endoscopic sphincterotomy. Pancreas. 2018;47(7):880-883.
- Gutta A, Fogel R, Sherman S. Identification and management of pancreas divisum. Expert Rev Gastroenterol Hepatol. 2019;13(11):1089-1105.
- Smanio T. Proposed nomenclature and classification of the human pancreatic ducts and duodenal papillae. Study based on 200 post mortems. Int Surg. 1969;52(2):125-141.
- Stimec B, Bulajic M, Korneti V, et al. Ductal morphometry of ventral pancreas in pancreas divisum. Comparison between clinical and anatomical results. Ital J Gastroenterol. 1996;28(2):76-80.
- Hernandez LV, Catalano MF. Endoscopic techniques (ERCP, EUS) for the evaluation of unexplained acute pancreatitis. TICE. 2004;6(3):84-90.
- Fogel EL, Toth TG, Lehman GA, et al. Does endoscopic therapy favorably affect the outcome of patients who have recurrent acute pancreatitis and pancreas divisum? Pancreas. 2007;34(1):21-45.
- Choudari CP, Imperiale TF, Sherman S, et al. Risk of pancreatitis with mutation of the cystic fibrosis gene. Am J Gastroenterol. 2004;99(7):1358-1363.
- Garg PK, Khajuria R, Kabra M, et al. Association of SPINK1 gene mutation and CFTR gene polymorphisms in patients with pancreas divisum presenting with idiopathic pancreatitis. J Clin Gastroenterol. 2009;43(9):848-852.
- Bertin C, Pelletier AL, Vullierme MP, et al. Pancreas divisum is not a cause of pancreatitis by itself but acts as a partner of genetic mutations. Am J Gastroenterol. 2012;107(2):311-317.
- Lin TK, Abu-El-Haija M, Nathan JD, et al. Pancreas divisum in pediatric acute recurrent and chronic pancreatitis: report from INSPPIRE. J Clin Gastroenterol. 2019;53(6):e232-e238.
- Klein SD, Affronti JP. Pancreas divisum, an evidence-based review: part I, pathophysiology. Gastrointest Endosc. 2004;60(3):419-425.
- Madacsy T, Pallagi P, Maleth J. Cystic fibrosis of the pancreas: the role of CFTR channel in the regulation of intracellular Ca(2+) signaling and mitochondrial function in the exocrine pancreas. Front Physiol. 2018;9:1585.
- Mosler P, Akisik F, Sandrasegaran K, et al. Accuracy of magnetic resonance cholangiopancreatography in the diagnosis of pancreas divisum. Dig Dis Sci. 2012;57(1):170-174.
- Shen Z, Munker S, Zhou B, et al. The accuracies of diagnosing pancreas divisum by magnetic resonance cholangiopancreatography and endoscopic ultrasound: a systematic review and meta-analysis. Sci Rep. 2016;6:35389.
- Mao Y, Ni J, Peng K, et al. Superiority of linear-array EUS over MRCP in diagnosing pancreas divisum: evidence from a multicenter retrospective study in Oriental cohorts (with video). Gastrointest Endosc. 2025;102(2):233-241.
- Lans JI, Geenen JE, Johanson JF, Hogan WJ. Endoscopic therapy in patients with pancreas divisum and acute pancreatitis: a prospective, randomized, controlled clinical trial. Gastrointest Endosc. 1992;38(4):430-434.
- Chacko LN, Chen YK, Shah RJ. Clinical outcomes and nonendoscopic interventions after minor papilla endotherapy in patients with symptomatic pancreas divisum. Gastrointest Endosc. 2008;68(4):667-673.
- Romagnuolo J, Durkalski V, Fogel EL, et al. Outcomes after minor papilla endoscopic sphincterotomy (MPES) for unexplained acute pancreatitis and pancreas divisum: final results of the multicenter prospective FRAMES (frequency of recurrent acute pancreatitis after minor papilla endoscopic sphincterotomy) study. Gastrointest Endosc. 2013;77(5 suppl):AB379.
- Zator ZA, Das R, Mounzer R, et al. Which patients with recurrent acute pancreatitis in the setting of pancreas divisum respond to minor sphincterotomy? Gastrointest Endosc. 2016;83(5):AB275-276.
- Schepis T, Pafundi PC, Tringali A, et al. Endoscopic minor papilla sphincterotomy in patients with complete pancreas divisum and acute recurrent pancreatitis: a metanalysis. Scand J Gastroenterol. 2024;59(2):225-231.
- Borak GD, Romagnuolo J, Alsolaiman M, et al. Long-term clinical outcomes after endoscopic minor papilla therapy in symptomatic patients with pancreas divisum. Pancreas. 2009;38(8):903-906.
- Mariani A, Di Leo M, Petrone MC, et al. Outcome of endotherapy for pancreas divisum in patients with acute recurrent pancreatitis. World J Gastroenterol. 2014;20(46):17468-17475.
- Tringali A, Voiosu T, Schepis T, et al. Pancreas divisum and recurrent pancreatitis: long-term results of minor papilla sphincterotomy. Scand J Gastroenterol. 2019;54(3):359-364.
- de Jong DM, Stassen PM, Poley JW, et al. Clinical outcome of endoscopic therapy in patients with symptomatic pancreas divisum: a Dutch cohort study. Endosc Int Open. 2021;9(7):E1164-E1170.
- Kwan V, Loh SM, Walsh PR, et al. Minor papilla sphincterotomy for pancreatitis due to pancreas divisum. ANZ J Surg. 2008;78(4):257-261.
- Kozarek RA. Pancreatic stents can induce ductal changes consistent with chronic pancreatitis. Gastrointest Endosc. 1990;36(2):93-95.
- Rashdan A, Fogel EL, McHenry L Jr, et al. Improved stent characteristics for prophylaxis of post-ERCP pancreatitis. Clin Gastroenterol Hepatol. 2004;2(4):322-329.
- Lehman GA, Sherman S. Diagnosis and therapy of pancreas divisum. Gastrointest Endosc Clin N Am. 1998;8(1):55-77.
- Cote GA, Durkalski-Mauldin VL, Serrano J, et al. SpHincterotomy for acute recurrent pancreatitis randomized trial: rationale, methodology, and potential implications. Pancreas. 2019;48(8):1061-1067.
- Cote GA, Durkalski-Mauldin V, Fogel EL, et al. Minor papilla sphincterotomy does not reduce the risk of acute pancreatitis in patients with acute recurrent pancreatitis and pancreas divisum: the SHARP trial. Digestive Disease Week 2025. Abstract 987a.
- Todani TY, Watanabe Y, Narusue M, et al. Congenital bile duct cysts: classification, operative procedures, and review of thirty-seven cases including cancer arising from choledochal cyst. Am J Surg. 1977;134(2):263-269.
- Sarris GE, Tsang D. Choledochocele:case report, literature review, and a proposed classification. Surgery. 1989;105(3):408-414.
- Venu RP, Geenen JE, Hogan WJ, et al. Role of endoscopic retrograde cholangiopancreatography in the diagnosis and treatment of choledochocele. Gastroenterology. 1984;87(5):1144-1149.
- Kim MH, Myung SJ, Lee SK, et al. Ballooning of the papilla during contrast injection: the semaphore of a choledochocele. Gastrointest Endosc. 1998;48(3):258-262.
- Law RM, Topazian M. Diagnosis and treatment of choledochoceles. Clin Gastroenterol Hepatol. 2014;12(2):196-203.
- Saleem NS. Unexplained acute pancreatitis and acute recurrent pancreatitis. In: Baron TH, Kozarek RA, Carr-Locke DL, et al, eds. ERCP. Elsevier; 2024:547-559.
- Sherman S. Choledochal cysts. Snape WJ, ed. In: Consultations in Gastroenterology. WB Saunders; 1996:814-828.
- Goldberg PB, Long WB, Oleaga JA, et al. Choledochocele as a cause of recurrent pancreatitis. Gastroenterology. 1980;78(5 pt 1):1041-1045.
- Park DH, Kim MH, Lee SK, et al. Can MRCP replace the diagnostic role of ERCP for patients with choledochal cysts? Gastrointest Endosc. 2005;62(3):360-366.
- Tio TL, Rohde P, Sie LH, et al. Endosonography in the pre-operative diagnosis of choledochocele. Gastrointest Endosc. 1992;38(3):381-383.
- Okamoto T, Fukuda K. Endosonographic diagnosis of choledochocele. Endosc Int Open. 2021;9(10):E1579-E1580.
- Martin RF, Biber BP, Bosco JJ, et al. Symptomatic choledochoceles in adults. Endoscopic retrograde cholangiopancreatography recognition and management. Arch Surg. 1992;127(5):536-538; discussion 538-539.
- Ladas SD, Katsogridakis I, Tassios P, et al. Choledochocele, an overlooked diagnosis: report of 15 cases and review of 56 published reports from 1984 to 1992. Endoscopy. 1995;27:233-239.
- Akkiz H, Colakoglu SO, Ergun Y, et al. Endoscopic retrograde cholangiopancreatography in the diagnosis and management of choledochal cysts. HPB Surg. 1997;10(4):211-218; discussion 218-219.
- Jang JY, Yoon CH, Kim KM. Endoscopic retrograde cholangiopancreatography in pancreatic and biliary tract disease in Korean children. World J Gastroenterol. 2010;16(4):490-495.
- Samavedy R, Sherman S, Lehman GA. Endoscopic therapy in anomalous pancreatobiliary duct junction. Gastrointest Endosc. 1999;50(5):623-627.
- Levy MJ, Geenen JE. Idiopathic acute recurrent pancreatitis. Am J Gastroenterol. 2001;96(9):2540-2555.
- Shioyama E, Mitoro A, Ogawa H, et al. A pancreatic mucinous cystic neoplasm undergoing intriguing morphological changes over time and associated with recurrent pancreatitis: a case report. Medicine (Baltimore). 2019;98(28):e16435.
- Muniraj T, Aslanian HR, Laine L, et al. Resection of pancreatic cystic neoplasms in recurrent acute pancreatitis prevents recurrent pancreatitis but does not identify more malignancies. World J Gastroenterol. 2021;27(15):1630-1642.
- Pitakteerabundit T, Fagenholz PJ, Luckhurst CM, et al. Acute pancreatitis in intraductal papillary mucinous neoplasm: associations with epithelial subtype, degree of dysplasia, and risk of recurrence. Surgery. 2025;181:109038.
- Elta GH, Enestvedt BK, Sauer BG, et al. ACG clinical guideline: diagnosis and management of pancreatic cysts. Am J Gastroenterol. 2018;113(4):464-479.
- Ohtsuka T, Fernandez-Del Castillo C, Furukawa T, et al. International evidence-based Kyoto guidelines for the management of intraductal papillary mucinous neoplasm of the pancreas. Pancreatology. 2024;24(2):255-270.
- Farrell JJ, Brugge WR. Intraductal papillary mucinous tumor of the pancreas. Gastrointest Endosc. 2002;55(6):701-714.
- Siddiqui AA, Shahid H, Shah A, et al. High risk of acute pancreatitis after endoscopic ultrasound-guided fine needle aspiration of side branch intraductal papillary mucinous neoplasms. Endosc Ultrasound. 2015;4(2):109-114.
- Kirkegård J, Gaber C, Lund JL, et al. Acute pancreatitis as an early marker of pancreatic cancer and cancer stage, treatment, and prognosis. Cancer Epidemiol. 2020;64:101647.
- Kim MH, Lee SK, Seo DW, et al. Tumors of the major duodenal papilla. Gastrointest Endosc. 2001;54(5):609-620.
- Nguyen N, Shah JN, Binmoeller KF. Outcomes of endoscopic papillectomy in elderly patients with ampullary adenoma or early carcinoma. Endoscopy. 2010;42(11):975-977.
- El Hajj II, Coté GA. Endoscopic diagnosis and management of ampullary lesions. Gastrointest Endosc Clin N Am. 2013;23(1):95-109.
- Ridtitid W, Tan D, Schmidt SE, et al. Endoscopic papillectomy: risk factors for incomplete resection and recurrence during long-term follow-up. Gastrointest Endosc. 2014;79(2):289-296.
- Pitlick MM, Abeykoon JP, Dao LN, et al. Non-Hodgkin lymphoma presenting as acute pancreatitis: a rare occurrence. Clin Case Rep. 2018;7(1):107-109.
- Lee M, Song JS, Hong SM, et al. Sarcoma metastasis to the pancreas: experience at a single institution. J Pathol Transl Med. 2020;54(3):220-227.
- El Jurdi K, Taleb A, Choucair K, et al. An unusual case of urothelial cell carcinoma with metastasis to the pancreas. Cureus. 2021;13(5):e14851.
- DeWitt J, Devereaux B, Chriswell M, et al. Comparison of endoscopic ultrasonography and multidetector computed tomography for detecting and staging pancreatic cancer. Ann Intern Med. 2004;141(10):753-763.
- Kuwatani M, Nakamura T, Hayashi T, et al. Clinical outcomes of biliary drainage during a neoadjuvant therapy for pancreatic cancer: metal versus plastic stents. Gut Liver. 2020;14(2):269-273.
- Cavestro GM, Zuppardo RA, Bertolini S, et al. Connections between genetics and clinical data: role of MCP-1, CFTR, and SPINK-1 in the setting of acute, acute recurrent, and chronic pancreatitis. Am J Gastroenterol. 2010;105(1):199-206.
- Jalaly NY, Moran RA, Fargahi F, et al. An evaluation of factors associated with pathogenic PRSS1, SPINK1, CTFR, and/or CTRC genetic variants in patients with idiopathic pancreatitis. Am J Gastroenterol. 2017;112(8):1320-1329.
- Whitcomb DC, Gorry MC, Preston RA, et al. Hereditary pancreatitis is caused by a mutation in the cationic trypsinogen gene. Nat Genet. 1996;14(2):141-145.
- Giefer MJ, Lowe ME, Werlin SL, et al. Early-onset acute recurrent and chronic pancreatitis is associated with PRSS1 or CTRC gene mutations. J Pediatr. 2017;186:95-100.
- Liu QY, Abu-El-Haija M, Husain SZ, et al. Risk factors for rapid progression from acute recurrent to chronic pancreatitis in children: report from INSPPIRE. J Pediatr Gastroenterol Nutr. 2019;69(2):206-211.
- Aoun E, Muddana V, Papachristou GI, et al. SPINK1 N34S is strongly associated with recurrent acute pancreatitis but is not a risk factor for the first or sentinel acute pancreatitis event. Am J Gastroenterol. 2010;105(2):446-451.
- Pelletier AL, Bienvenu T, Rebours V, et al. CFTR gene mutation in patients with apparently idiopathic pancreatitis: lack of phenotype-genotype correlation. Pancreatology. 2010;10(2-3):158-164.
- LaRusch J, Lozano-Leon A, Stello K, et al. The Common Chymotrypsinogen C (CTRC) Variant G60G (C.180T) Increases Risk of Chronic Pancreatitis But Not Recurrent Acute Pancreatitis in a North American Population. Clin Transl Gastroenterol. 2015;6(1):e68.
- Culetto A, Bournet B, Haennig A, et al. Prospective evaluation of the aetiological profile of acute pancreatitis in young adult patients. Dig Liver Dis. 2015;47(7):584-589.
- Joergensen M, Brusgaard K, Cruger DG, et al. Incidence, etiology and prognosis of first-time acute pancreatitis in young patients:a population-based cohort study. Pancreatology. 2010;10(4):453-461.
- Phadke MY, Sellers ZM. Current clinical opinion on CFTR dysfunction and patient risk of pancreatitis: diagnostic and therapeutic considerations. Expert Rev Gastroenterol Hepatol. 2022;16(6):499-509.
- Whitcomb DC. PRSS1-Related Hereditary Pancreatitis. In: Adam MP, Feldman J, Mirzaa GM, et al, eds. GeneReviews. University of Washington; 1993.
- Madhani K, Farrell JJ. Autoimmune pancreatitis: an update on diagnosis and management. Gastroenterol Clin North Am. 2016;45(1):29-43.
- Chari ST, Smyrk TC, Levy MJ, et al. Diagnosis of autoimmune pancreatitis: the Mayo Clinic experience. Clin Gastroenterol Hepatol. 2006;4(8):1010-1016; quiz 1934.
- Okazaki K, Kawa S, Kamisawa T, et al. Clinical diagnostic criteria of autoimmune pancreatitis: revised proposal. J Gastroenterol. 2006;41(7):626-631.
- Shimosegawa T, Chari ST, Frulloni L, et al. International consensus diagnostic criteria for autoimmune pancreatitis: guidelines of the International Association of Pancreatology. Pancreas. 2011;40(3):352-358.
- Zhang L, Chari S, Smyrk TC, et al. Autoimmune pancreatitis (AIP) type 1 and type 2: an international consensus study on histopathologic diagnostic criteria. Pancreas. 2011;40(8):1172-1179.
- Ha J, Choi SH, Byun JH, et al. Meta-analysis of CT and MRI for differentiation of autoimmune pancreatitis from pancreatic adenocarcinoma. Eur Radiol. 2021;31(5):3427-3438.
- Palazzo M, Palazzo L, Aubert A, et al. Irregular narrowing of the main pancreatic duct in association with a wall thickening is a key sign at endoscopic ultrasonography for the diagnosis of autoimmune pancreatitis. Pancreas. 2015;44(2):211-215.
- Kamisawa T, Chari ST, Giday SA, et al. Clinical profile of autoimmune pancreatitis and its histological subtypes: an international multicenter survey. Pancreas. 2011;40(6):809-814.
- Gallo C, Dispinzieri G, Zucchini N, et al. Autoimmune pancreatitis: cornerstones and future perspectives. World J Gastroenterol. 2024;30(8):817-832.
- Levy MJ, Reddy RP, Wiersema MJ, et al. EUS-guided trucut biopsy in establishing autoimmune pancreatitis as the cause of obstructive jaundice. Gastrointest Endosc. 2005;61(3):467-472.
- Moon SH, Kim MH. The role of endoscopy in the diagnosis of autoimmune pancreatitis. Gastrointest Endosc. 2012;76(3):645-656.
- Moon SH, Kim MH, Park DH, et al. IgG4 immunostaining of duodenal papillary biopsy specimens may be useful for supporting a diagnosis of autoimmune pancreatitis. Gastrointest Endosc. 2010;71(6):960-966.
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Management of Idiopathic Acute Pancreatitis And Recurrent Acute Pancreatitis
Management of Idiopathic Acute Pancreatitis And Recurrent Acute Pancreatitis