Co-Director, Developmental Endoscopy Unit
Mayo Clinic
Rochester, Minnesota
Endoscopic ultrasound has evolved from a purely diagnostic modality into a powerful therapeutic tool in interventional gastroenterology. Therapeutic EUS is increasingly used for a wide range of procedures, including fine-needle biopsy, drainage of pancreatic and postoperative fluid collections, biliary access and drainage, gastroenterostomy, and novel nonsurgical bypass procedures, among others.
Earlier this year, the American Society for Gastrointestinal Endoscopy published quality indicators for EUS that included ideal adverse event rate thresholds for diagnostic and therapeutic EUS procedures.1 This guidance aims to standardize the rapidly evolving practice of EUS by minimizing complications and ensuring high-quality gastrointestinal patient care through use of quality indicators, as well as EUS report standards, training and competency criteria, and a framework for multidisciplinary collaboration.
Although these efforts are critically important for improving the safety and efficacy of EUS procedures worldwide, the design and functionality of the echoendoscope is another increasingly recognized factor in the successful and safe performance of EUS procedures. As therapeutic applications continue to expand, the importance of therapeutic echoendoscope design has become central to the efficacy, accessibility, and safety of EUS-guided interventions. Below, I explore several ideas related to how echoendoscope design affects therapeutic EUS procedures and why innovation in this area is essential for the future of minimally invasive EUS-guided gastroenterology procedures.
Evolution of EUS and the Role Of Endoscope Design
EUS initially was developed to enhance diagnostic imaging of the gastrointestinal tract, providing high- resolution visualization of the digestive organs and surrounding tissues. Traditional linear echoendoscopes offered limited maneuverability and working channel access, which restricted therapeutic capabilities mainly to EUS-guided fine-needle aspiration (FNA) for sampling and injection. As interventional EUS procedures have gained popularity and the number of available devices to support these interventions has grown, the limitations of early scope design have become apparent. The need for more robust tools capable of combining precision imaging with therapeutic functionality has led to significant innovations in endoscope architecture, including some interesting new platforms through which endoscopists can offer EUS procedures in their practices.
Scope Design Affects Therapeutic Precision and Safety
Precision and safety are 2 pillars of a successful therapeutic EUS practice, and advanced endoscope designs contribute significantly to both. First, improved maneuverability—specifically, better articulation and control—enables operators to access difficult-to-reach anatomic locations. Enhanced tip deflection and elevator mechanisms allow the endoscopist to align instruments more accurately with target lesions or ducts, reducing procedural time and minimizing trauma from repeated needle or device advancements.
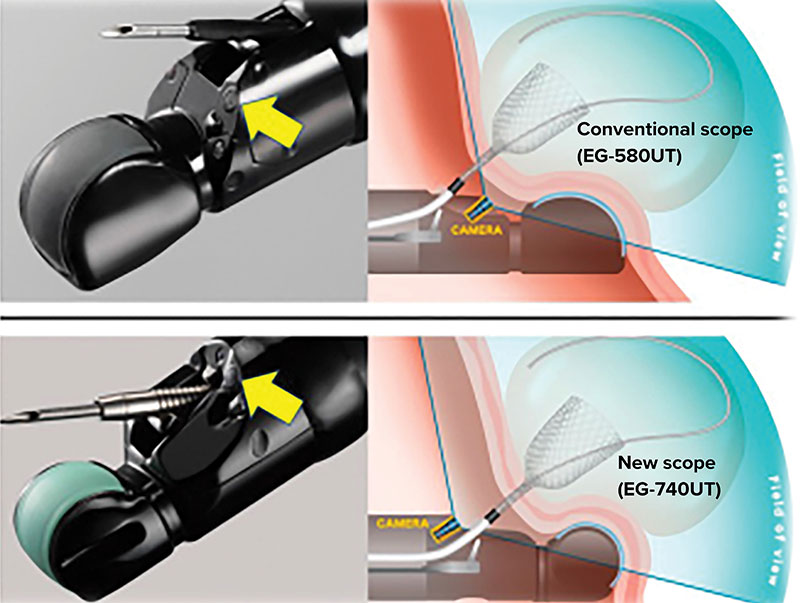
Fujisawa et al describe significant improvements in procedural and fluoroscopy time for therapeutic EUS cases using a novel interventional EUS scope design in a study that included 143 procedures in 120 patients.2 Their study compared procedures using an older conventional model (EG-580UT, Fujifilm) with a newer scope design (EG-740UT, Fujifilm). The newer model includes a bending segment curvature that improved from 27.9 to 25 degrees; enhanced elevator strength, allowing for an impressive increased angle of deflection from 35.2 to 76.7 degrees (Figure 1); and a white light endoscopy camera that was relocated from distal to the working channel to a proximal location, allowing for better visualization of tools being deployed via the working channel (Figure 2).
From Our Case Files: ERCP With an EUS Scope
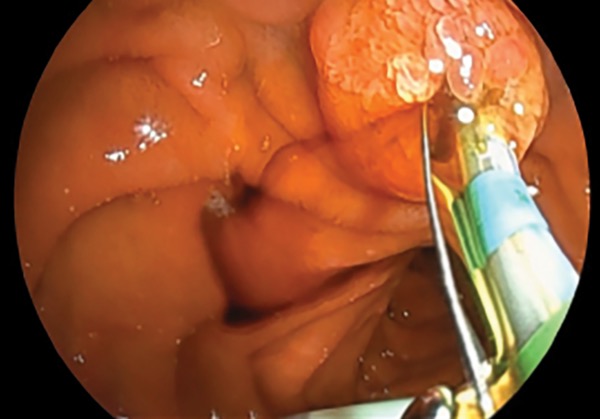
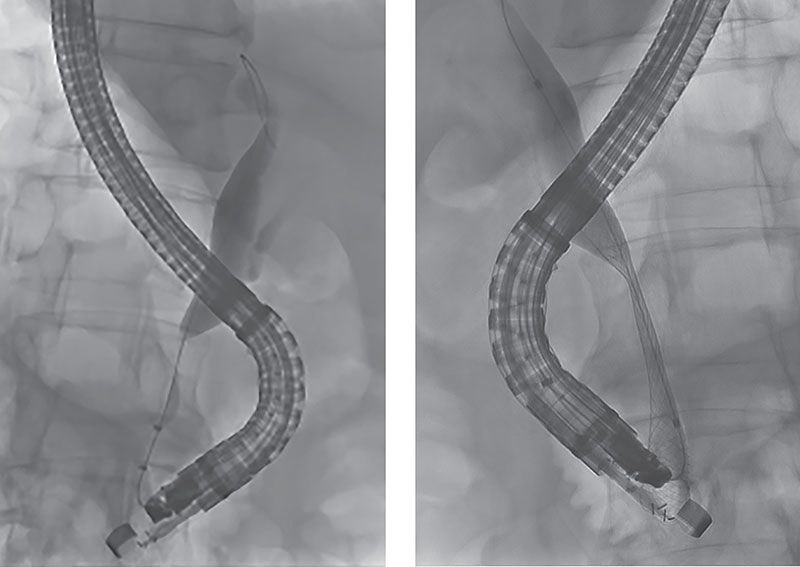
A 64-year-old man presented with jaundice, biliary dilation, and a new mass lesion in the head of the pancreas. EUS was performed with the EG-740UT scope for staging and sampling of the primary mass lesion, which confirmed pancreatic adenocarcinoma. Using the same scope, the endoscopist was able to perform transpapillary endoscopic retrograde cholangiopancreatography (ERCP) due to the strength and deflection of the elevator and view of the accessory channel allowed by this new scope design (Figure 3). A fully covered metal stent was deployed across the distal malignant biliary obstruction for palliation of the stricture after biliary sphincterotomy (Figure 4).
Other recent EUS scope design features include the integration of high-frequency ultrasound probes with Doppler capabilities, which aids in identifying vascular structures and avoiding inadvertent vessel puncture. Also, innovations in imaging fidelity, such as contrast-enhanced EUS and elastography, are being integrated into many echoendoscope processor platforms, offering even more detailed assessments before intervention.
Furthermore, the development of varying diameters and rigidity in echoendoscopes can affect patient comfort, navigation ease, and procedural outcomes. Slimmer, more flexible scopes reduce the need for deep sedation or general anesthesia and may reduce the risk for complications, particularly in frail or smaller patients. Bang et al show that a slim linear echoendoscope significantly improved their practice of interventional and diagnostic EUS (Figure 5).3 In their reported experience of more than 2,000 procedures, complete EUS examination failed in 1.15% of cases (n=23) due to the diameter of the EUS scope. Complete examination was technically successful in 22 of those 23 failed cases (>95% successful rescue) using a slim EUS scope design (EG34-J10U, Pentax). This further highlights the role of scope design in improving the diagnostic and therapeutic capabilities of physicians working on the frontiers of EUS.
From Our Case Files: Endobronchial Ultrasound Scope for GI Access
A 2-year-old girl presented with failure to thrive and vomiting after meals. Evaluation included CT, which revealed a mass lesion in the pancreas. Percutaneous sampling was unsuccessful, so EUS-guided tissue sampling was requested to guide next steps in therapy. However, the patient’s weight of 10 kg made passage of an adult linear echoendoscope, which has an insertion tube diameter of 12.6 mm, challenging and ill-advised. Thus, we opted to use an endobronchial ultrasound (EBUS) scope (BF-UC180F, Olympus) with a 6.2-mm insertion shaft diameter. The imaging field with EBUS is smaller and quality of the image is reduced compared with larger EUS scopes (Figure 6).
A solid cystic lesion was seen in the neck of the pancreas, which was sampled using EBUS FNA. Final cytology from the sampling was consistent with pancreatoblastoma, which was resected with no evidence of tumor recurrence. This case serves as a reminder that “bigger” is not always “better,” and EUS scope platforms should consider smaller and abnormal anatomy, where a slim-scope design may permit life-changing diagnostic and therapeutic interventions.
Facilitating Access to EUS And Reducing the Learning Curve
Therapeutic EUS remains a technically demanding skill that requires extensive training and experience. Poor scope design can compound difficulties that practicing physicians may take for granted. These shortfalls in design likely increase the learning curve and raise the risk for complications in not only novice but also expert hands. User-friendly scope design with built-in guidance and safety mechanisms likely will be essential to democratize access to EUS across more centers and geographic regions.
Simplified control systems, visual feedback related to tip orientation within a patient’s anatomy, and intuitive interfaces and image interpretation software already are being explored for future-generation endoscopes. Future designs might include real-time haptic feedback or automated safety locking systems to prevent failed access, unintended puncture, or stent maldeployment. These features improve training and help maintain consistency and safety among less experienced practitioners.
Another limitation to improved access to EUS for patients across the globe is scope and processor cost, which may be prohibitive to new, smaller, or rural practices. In response to this issue, a novel platform called the EndoSound Vision System (EndoSound) is available in the United States as an add-on device that converts any flexible upper gastrointestinal video endoscope and most pediatric colonoscopes into a functional EUS system (Figure 7). This system is comprised of a compact ultrasound beamformer, reusable transducer, and disposable mounting kit and allows for advanced procedures, such as fine-needle biopsy, without the need for the elevator mechanism used in traditional reusable linear echoendoscopes. This system could improve access to safe and efficient EUS diagnostic and interventional procedures where expertise is available but access to modern EUS equipment is lacking.

Finally, simulation-based training through virtual reality and simulated phantom (physical) models is becoming an integral part of many fields within medical education. Realistic, low-risk training opportunities are critical for developing the next generation of procedural providers who can keep up with the growing field of diagnostic and therapeutic EUS. Echoendoscopes that can be easily integrated with simulators and training devices could become critical for more realistic training, practice, and assessment of current and future EUS procedures, ultimately accelerating procedural proficiency and improving patient experiences and outcomes.
Toward a Future of Robotic, AI-Guided, And Personalized Medical Interventions
As therapeutic EUS continues to push boundaries—enabling procedures that were once the exclusive domain of surgery—the need for precision grows. Design elements of the future may include multiple accessory ports, enhanced water jet systems for clearing the field of view, and ergonomic control interfaces, such as potential integration with robotic-assisted technologies or motorized insertion systems. Robotic assistance may one day allow for even more accurate, less operator-dependent interventions aimed at further empowering the endoscopists of the future to perform high-end interventional procedures with confidence.
In bronchoscopy, peripheral lung lesion assessment has been revolutionized by a robotic bronchoscopy platform (Ion, Intuitive Surgical), made possible through shape-sensing fibers, an ultrathin access catheter, and highly precise robotic control.4
The intersection of endoscope design with artificial intelligence is another area poised for evolution in echoendoscope design. AI-enhanced EUS systems—those that can provide real-time feedback, recognize anatomic landmarks, and recommend diagnoses and/or procedural steps—will depend heavily on advanced data processing in development.5 The form factor and internal architecture of these future echoendoscopes must be engineered to support these improvements without compromising usability or safety, such as factors important to natural orifice translumenal endoscopic surgery, ensuring sterility.
Furthermore, personalized medicine in EUS may someday require the ability to tailor instruments to individual patient anatomies and pathologies. Endoscope platforms with interchangeable working shafts, variable stiffness, and more complex patient-specific pre-procedural and intra-procedural planning for diagnostic and therapeutic intents via 3D EUS (or dual cross-sectional modality) mapping will become more desirable. These goals necessitate rethinking our current scope designs, which were engineered as diagnostic-only tools, as the future likely will require more versatile, adaptable platforms.
Conclusion
EUS has evolved from a diagnostic imaging modality to a keystone platform for therapeutic intervention in flexible endoscopy and gastroenterology. The future of the field hinges not only on procedural and device innovations but also on the scope platforms that make them possible. Therapeutic EUS has grown to encompass a wide array of procedures, from earlier biopsy and nerve blocks to newer and more advanced novel anastomosis creation and ablation procedures. The success and safety of these interventions may be intricately linked to echoendoscopic equipment design. As the scope of therapeutic intent on the frontiers of EUS expands, it is difficult to overstate the importance of innovative design.
Advanced scope designs can enhance safety, expand therapeutic options, reduce the learning curve, and may pave the way for robotic and AI integration. Newer EUS scope designs have involved significant improvements to the maneuverability, shaft size, camera view, and elevator function of therapeutic linear scopes. Early data suggest that procedure duration can be shortened by these improvements, and previously challenging interventions, including ERCP maneuvers, using an EUS scope are made simpler, as shown in our case examples.
Investment in therapeutic echoendoscope design and engineering should remain a strategic imperative for scope manufacturers and clinicians seeking to stay at the forefront of minimally invasive gastrointestinal care. In the coming decade, as therapeutic EUS becomes even more central to the management of complex gastrointestinal and hepatobiliary conditions, these advances in echoendoscope technology from the frontiers of EUS will allow us to reach the full potential of minimally invasive endoscopic therapy.
References
- Mishra G, Lennon AM, Pausawasdi N, et al. Quality indicators for EUS. Gastrointest Endosc. 2025;101(5):928-949.
- Fujisawa T, Ishii S, Nakai Y, et al. Dedicated echoendoscope for interventional endoscopic ultrasound: comparison with a conventional echoendoscope. J Clin Med. 2024;13(10):2840.
- Bang JY, Willems P, Navaneethan U, et al. Impact of slim linear echoendoscope on the practice of EUS. J Clin Gastroenterol. 2024;58(8):830-835.
- Simoff MJ, Pritchett MA, Reisenauer JS, et al. Shape-sensing robotic-assisted bronchoscopy for pulmonary nodules: initial multicenter experience using the Ion™ Endoluminal System. BMC Pulm Med. 202116;21(1):322.
- Marya NB, Powers PD, Chari ST, et al. Utilisation of artificial intelligence for the development of an EUS-convolutional neural network model trained to enhance the diagnosis of autoimmune pancreatitis. Gut. 2021;70(7):1335-1344.
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() The Frontiers of Therapeutic Endoscopic Ultrasound in 2025: Advances in EUS Scope Design
The Frontiers of Therapeutic Endoscopic Ultrasound in 2025: Advances in EUS Scope Design