Houston, Texas
Wireless capsule endoscopy (WCE) has been transforming the field of gastroenterology since the early 2000s as a noninvasive method to visualize the gastrointestinal tract, particularly the small intestine, which traditionally has been challenging to examine.
The concept of WCE was first developed in Israel in 1981 by Gavriel Iddan, a military engineer, and Eitan Scapa, MD, a gastroenterologist.1-3 Meanwhile, in the United Kingdom, gastroenterologist Paul Swain, MD, succeeded in sending images from a pig’s stomach to a monitor2 and was the first human to swallow a capsule endoscope. These innovations brought the concept of “swallowing the surgeon,” proposed by Nobel Prize–winning theoretical physicist Richard Feynman decades earlier, closer to reality. WCE emerged as a solution to visualizing the small bowel completely, something traditional fiberoptic endoscopy could not do.2
WCE, originally known as the PillCam and manufactured by Given Imaging, received its first FDA approval in 2001.4 The PillCam SB1 measured 26×11 mm, had an 8-hour battery, and provided images with a resolution of 256×256 pixels.1,3 This capsule’s success quickly broadened applications of WCE from obscure GI bleeding to include assessment of Crohn’s disease, celiac disease, small-bowel tumors (SBTs), and iron deficiency anemia.5 Later innovations resulted in colon capsule endoscopy (CCE) and pan-enteric capsules, allowing visualization of both the small intestine and colon.6,7 As demand for noninvasive diagnostic options continues to grow, capsule endoscopy has evolved to include potential new uses, including colorectal cancer (CRC) screening.8
The integration of artificial intelligence (AI) into capsule endoscopy technology has improved diagnostic accuracy and efficiency, shortening image interpretation time while preserving high sensitivity for identifying mucosal abnormalities.9 WCE still encounters challenges such as incomplete examinations, capsule retention, and a lack of therapeutic functions,10 but ongoing advances in capsule robotics, active locomotion, magnetic control, and real-time lesion detection are poised to transform WCE from solely a diagnostic tool to an intervention-capable platform.11 This review covers capsule endoscopy’s current applications, technological advancements, limitations, and future prospects, emphasizing its critical role in the advancing field of GI diagnostics and therapeutics.
Technology
Hardware
Capsule design balances high-resolution imaging, efficient illumination, reliable communication, long battery life, and minimal invasiveness in a compact form, typically smaller than 32×12 mm.7,12 WCE systems include a capsule endoscope, a patient-worn sensing system (with sensing arrays or a belt), a data recorder with a battery pack, and software for reviewing and interpreting images.5 Real-time image review during exams is available with some systems. The capsule typically has a smooth, transparent, biocompatible outer shell, commonly polycarbonate, to reduce mucosal damage during transit.13 Early capsule endoscopes used a single optical dome and unidirectional camera, but modern designs incorporate 2 (or more) cameras for a wider field of view and more complete mucosal visualization (Table).7
The core imaging technology relies on complementary metal oxide semiconductor (CMOS) sensors, which replaced charge-coupled devices due to lower power consumption and smaller size.2 The development of CMOS was a critical breakthrough that enabled the first successful human trials with adequate resolution (about 256×256 pixels) while using much less power.3 Illumination is provided by energy-efficient light-emitting diodes (LEDs), which deliver high-intensity, low-heat lighting that is ideal for the GI tract’s dark conditions.2,3 Synchronizing LEDs with image capture and transmission cycles conserves battery life, supporting extended recording sessions.5 Power typically is supplied by silver oxide or lithium ion button batteries, with early models offering about 8 hours of operation, sufficient for small-bowel transit.5 Advanced capsules, such as the OMOM Smart Capsule (Jinshan) and CapsoCam Plus (Capsovision), extend battery life up to 15 hours, enabling comprehensive small-bowel and pan-enteric imaging.7,14
Wireless transmission varies by manufacturer. Early systems used ultra–high-frequency radio telemetry, whereas newer models, such as the MiroCam (IntroMedic), employ human body communication, transmitting data through the patient’s tissues for improved reliability and reduced signal loss.3,5,11 On-board electronics as well as application-specific integrated circuits manage sensor operation, data compression, and transmission and are optimized for minimal energy use.3
Recent innovations such as the PillCam Genius SB (Medtronic) and OMOM HD (Jinshan) incorporate AI processors in or near the capsule’s receiving unit, enabling automated abnormality detection, rapid image triage, and reduced reading time.9,15 Magnetically controlled capsule (MCE) endoscopes, such as the NaviCam MCCE (AnX Robotica), use embedded magnets guided by external magnetic fields to enhance maneuverability and diagnostic accuracy in the stomach.16
The first CCE device was introduced in 2006 by Given Imaging as a minimally invasive technique for imaging of the colon that did not require sedation or gas insufflation.17 Due to the first generation’s inadequate accuracy, a second-generation CCE, PillCam COLON 2 (Medtronic), was developed.18 This capsule features a 172-degree viewing angle (nearly 360 degrees combined) and an adaptive frame rate function, capturing more images (35 frames per second) when moving faster and fewer (4 frames per second) when moving more slowly.18
Software
Images captured by WCEs are transmitted wirelessly to an external recorder or stored on board for later download, depending on the make and model.19 These data then are processed and displayed as video at user-selectable rates, typically from 3 to 40 frames per second.5 Software can help identify items of interest, for example, using red pixel detection in blood indicator algorithms to aid in identifying bleeding lesions.20,21 Capsule software can also track passage of the capsule through the GI tract, calculate gastric and small bowel transit times, capture and annotate images, and generate reports. Some programs have integrated quick-reference image atlases.5,22 Adaptive frame rate technology enables the capsule to take images at a faster rate during rapid movement and a slower rate during slower movement, optimizing image capture and preserving battery life.7,14
Clinical Applications
Obscure GI Bleeding
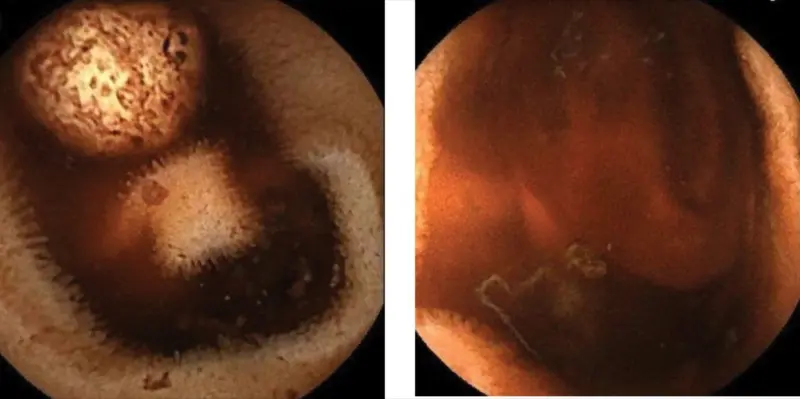
WCE can detect small-bowel bleeding in approximately 50% to 55% of cases with obscure GI bleeding in which upper and lower endoscopies are negative (Figures 1A and 1B).23 Also, studies have shown a negative predictive value as high as 97% when no significant abnormalities are found in the small bowel (Figure 2).24,25 However, its effectiveness is time-sensitive. The diagnostic yield is around 80% when performed within 48 hours from the last bleeding episode, but it drops significantly when performed more than 72 hours later.23 While evidence for the optimal timing in suspected occult small-bowel bleeding is less clear, the principle of performing WCE as soon as possible applies in cases with obscure GI bleeding.23
Crohn’s Disease
Approximately two-thirds of patients with Crohn’s disease have small-bowel involvement, frequently in the terminal ileum (~90%), where disease activity may be uneven, possibly resulting in false-negative findings on ileocolonoscopy.5 WCE can detect ulcers, erosions, erythema, and strictures in patients with Crohn’s disease,26 and it can be especially valuable for aggressive proximal or mid–small-bowel Crohn’s disease that cannot be reached during ileocolonoscopy.5 WCE provides a diagnostic yield comparable to that of magnetic resonance enterography (MRE) and colonoscopy in non-stricturing Crohn’s disease.27
WCE serves as a stand-alone diagnostic or monitoring tool in Crohn’s disease due to its ability to assess the entire digestive tract.27 Standardized quantitative scoring systems, such as the Capsule Endoscopy Crohn’s Disease Activity Index and Lewis score, are used to characterize the type, location, and severity of small-bowel lesions seen with WCE in patients with Crohn’s disease.28,29 Endoscopic recurrence of Crohn’s disease in the neo-terminal ileum after surgical resection is frequent, and WCE demonstrates a strong correlation with ileocolonoscopy in evaluating active disease in this region.30,31
Meckel’s Diverticulum
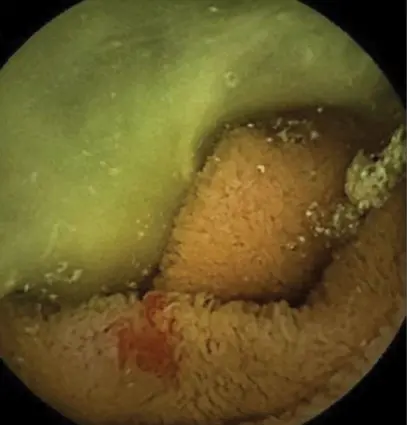
A recent systematic review of 33 case reports included 43 cases of Meckel’s diverticulum diagnosed with WCE.32 These patients, mostly young men (median age, 20 years), typically had an average of 3 negative tests, most commonly upper endoscopy, lower endoscopy, and Meckel’s scan, before undergoing WCE.32 The most frequent specific findings on WCE suggestive of Meckel’s diverticulum include the double lumen sign, a visible diverticulum entrance, and active bleeding (Figure 3).32
Small-Bowel Tumors
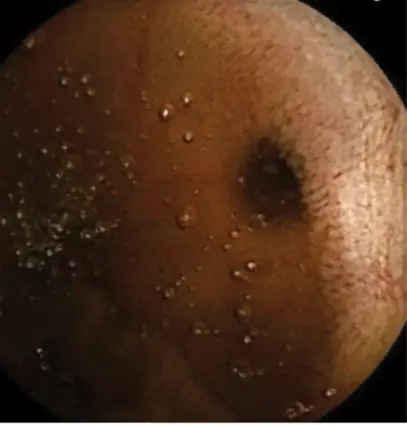
SBTs are uncommon, comprising just 2% of all GI tract cancers, and can be difficult to diagnose because they are frequently beyond the reach of standard scopes.20 WCE is valuable for detecting SBTs and polyps, particularly in patients with hereditary polyposis syndromes such as familial adenomatous polyposis and Peutz-Jeghers syndrome.4 However, SBTs can be challenging to detect because the color and texture of submucosal lesions may closely resemble the surrounding tissue (Figure 4).20 A pooled analysis found an 18.9% rate of missed tumor detections in capsule endoscopy studies.11 CT enterography (CTE) may be more sensitive than WCE when specifically looking for tumors, and CTE and MRE can provide additional extraluminal details that WCE cannot. Polyp and tumor size can affect the comparative sensitivity of WCE and MRE, with WCE seemingly more sensitive for smaller lesions.5
Celiac Disease
WCE is not recommended as a primary diagnostic modality for suspected celiac disease, except when a patient cannot undergo endoscopy and has a high pretest probability with positive serologies.5 However, WCE is a helpful supplementary tool for diagnosing celiac disease in equivocal cases and for monitoring complicated cases.
A systematic review of 22 studies found that when WCE was used to diagnose celiac disease in patients with negative serology or negative histology, it identified an endoscopic feature suggestive of celiac disease in about half of the cases.33 This is significant because histologic changes in celiac disease can be unevenly distributed and may be overlooked by duodenal biopsies alone. WCE captures numerous images of the entire small bowel, enhancing the likelihood of detecting a suggestive feature.33
Esophagogastroduodenoscopy with duodenal biopsies alone usually is insufficient to diagnose refractory celiac disease (RCD), and recent European guidelines suggest providing WCE as a complementary investigation to all patients with suspected or confirmed RCD to verify the diagnosis or identify complications of RCD, such as ulcerative jejunoileitis, enteropathy-associated T-cell lymphoma (EATL), and small-bowel adenocarcinoma.34
Barret et al used WCE to investigate whether it can predict the severity of celiac disease and detect complications in RCD. After tissue sampling via device-assisted enteroscopy, they identified 3 cases of EATL and 5 of ulcerative jejunoileitis.35 Atlas et al identified a tumor in the jejunum using WCE; subsequent biopsies revealed adenocarcinoma with regional lymph node metastases.36
Chronic or Functional Abdominal Pain
WCE can be helpful in patients with abdominal pain lasting 3 months or longer with no pathologic findings on upper endoscopy, colonoscopy, and/or imaging.34,37 Case reports and case series have described WCE detection of Meckel’s diverticulum, eosinophilic enteritis, and SBTs in patients with chronic unexplained abdominal pain.34 Retrospective studies have shown a low diagnostic yield, between 17% and 28%.38,39
Hereditary Gastrointestinal Polyposis
Capsule endoscopy is a safe and effective option for patients with polyposis syndromes, even in those who have had previous intestinal surgery.5 The American Society for Gastrointestinal Endoscopy recommends screening patients with familial adenomatous polyposis with stage IV or advanced duodenal polyposis for jejunal and ileal adenomas using either WCE or MRE, as these patients are at the highest risk.5 Vibrating capsule endoscopy is more sensitive for detecting small polyps (<5 mm) than MRE, has similar detection rates for polyps larger than 10 mm, and is more sensitive for polyps larger than 15 mm.5,34 A meta-analysis of 15 studies (821 patients) confirmed a high concordance (93%) between device-assisted enteroscopy and small-bowel capsule endoscopy in identifying small-bowel polyps and tumors.40
Monitoring Ulcerative Colitis
CCE, using the second-generation PillCam COLON 2 or pan-enteric capsules such as PillCam Crohn’s, has been used to assess disease activity and monitor mucosal healing in patients with ulcerative colitis.7,23 A systematic review and meta-analysis determined that CCE for ulcerative colitis had a pooled sensitivity of 92% and specificity of 71%.6 Another systematic review reported a pooled sensitivity rate of 93%, specificity at 68.8%, positive predictive value at 89.4%, and negative predictive value at 78.6% for detecting mucosal inflammation in ulcerative colitis.41
Capsule Scoring of Ulcerative Colitis, developed as an endoscopic severity score for the PillCam COLON 2, has shown a correlation with biomarkers and clinical scores similar to the Ulcerative Colitis Endoscopic Index of Severity.7,41 The score consists of 3 parameters: vascular pattern, bleeding, and erosions/ulcers.7 CCE provides a lower-quality image compared with conventional colonoscopy, and its visual presentation differs due to the lack of air and carbon dioxide insufflation.41
CRC Screening
CCE increasingly is being explored as a more environmentally friendly alternative to colonoscopy or a complementary tool to colonoscopy for CRC screening.18 It is noninvasive, does not require sedation or radiation, and is more comfortable for patients than colonoscopy.42 Some studies show it can be effectively performed in a community setting or even at home.43 A Danish CRC screening program found that there was no difference between CCE and colonoscopy in the detection rate of advanced neoplasia.44 A recent National Health Service study in England that included 10,369 patients concluded that CCE is likely to overdiagnose polyps, and, hence, colonoscopy should remain the gold-standard test.43
Limitations
Manual Review and Safety
WCE can produce anywhere between 50,000 and 80,000 images during its passage through the gastrointestinal tract.45 Analyzing and processing these images demand significant human resources. Even for skilled endoscopists, a thorough review can take at least 45 minutes.46 This intensive manual process is time-consuming and exhausting, raising the risk for missed or incorrect diagnoses.45 In addition, the anatomy of the digestive tract is complex and lesions vary in shape and characteristics across different regions, complicating image analysis. Small, irregularly shaped, or obscured lesions may be difficult to detect accurately, further increasing the likelihood of misdiagnosis.45 The capsule travels passively through the digestive tract, which may lead to constraints such as limited visibility and potentially missed lesions.47
Retention
The overall capsule retention rate (RR) has been reported to be between 0.73% and 2%. A 2017 systematic review of 33 studies consisting of 8,513 patients found a pooled RR of 2.1% of patients undergoing evaluation for small-bowel bleeding.48 Pooled RR was 3.6% in patients with suspected inflammatory bowel disease (IBD) and 8.2% for established IBD in this study. A 2020 meta-analysis of 108,079 vibrating capsule endoscopy procedures reported a pooled RR of 0.73%.49 In 2022, Cortegoso Valdivia et al reported an overall RR of 2% for all indications and 4% for Crohn’s disease in a meta-analysis of 328 original articles involving 86,930 patients.50
One large European single-center study of 2,401 patients found previous GI surgery significantly increased the risk for capsule retention, with an odds ratio of 7.64 (95% CI, 3.45-16.93; P<0.001).51 Capsule retention typically is asymptomatic, and a capsule may stay in the small bowel without causing issues for months or be expelled naturally during follow-up.52
A bowel obstruction rate of 1.9% was reported in a multicenter study from Spain in 2015.53 Retained capsule– causing symptoms may require surgery (46%-59% of cases), endoscopic management (13%-26%), or medical therapy in the form of bowel prep, enema, or nonsteroidal anti-inflammatory drugs (5%).49,54,55 Capsule retention in diverticula is extremely rare, with only 34 cases reported.10 Retention in diverticula, including Meckel’s diverticulum, mainly affects older male patients and often is asymptomatic (50% of reported cases). The longest reported case of retention was 7.5 years within Meckel’s diverticulum.56 Management of capsule retention in diverticula includes spontaneous resolution (20.6%), endoscopic treatment (35.3%), and surgical management (32.4%).10
Future Directions
Artificial Intelligence
In contrast with other endoscopy techniques that necessitate real-time interpretation and intervention, WCE is ideal for AI-based analysis, as it leverages pattern recognition across thousands of still images.20,57 Manually reviewing the large volume of images is time-intensive (30-120 minutes per video) and susceptible to human error, such as overlooking lesions.11,12,45 AI seeks to standardize image analysis, shorten review time, and enhance diagnostic accuracy.58,59
Convolutional neural networks (CNNs), deep learning algorithms in WCE software to analyze images, have been employed to improve detection of ulcers, erosions, angioectasia, and SBTs.9,60 CNNs have been demonstrated to decrease the time needed to review endoscopy images while maintaining detection accuracy.61 Computer-aided diagnosis, once dependent on traditional features and CNNs to extract high-level information from WCE images to enhance system performance, has been transformed by deep learning advances that have significantly improved the accuracy of diagnosis.62 A recent meta-analysis by Dhali et al found that AI-assisted WCE had diagnostic accuracy, sensitivity, and positive predictive values that were superior to those of standard capsule endoscopy.63
The FDA approved the first AI-assisted reading system for commercial use in 2024, the NaviCam small-bowel system (Ankon) with ProScan deep neural network (AnX Robotica).9 The diagnostic yield of small-bowel bleeding lesions was superior in AI-assisted reading compared with standard reading, and mean small-bowel reading time was 3 to 8 minutes using AI versus 33 to 37 minutes with standard reading.9 These differences were statistically significant. AI algorithms are being developed for automated detection and characterization of lesions; polyp detection, segmentation, and size estimation; diagnosis of specific conditions (polyps, epithelial tumors, submucosal tumors, nodules, ulcers, erosion, melanosis, venous structures, and others); image quality assessment; and localization and transit time estimation.21,45,62-66
Role for Medication Delivery
Passive drug delivery systems. Passive systems move the drug from capsule to the gut by natural diffusion.12 Early attempts included the high-frequency capsule from the 1980s, which used an external radiofrequency signal to deploy a controlled bolus of ciprofloxacin at selected sites in the GI tract.67 RaniPill (Rani Therapeutics) worked by inflating a balloon that would expose a dissolvable needle to inject a drug in the small bowel.68 Although passive delivery systems are appealing, they can fail if electromagnetic signals are attenuated by tissue or if the enclosure is not perfectly sealed, which can lead to drug leakage.12
Active drug delivery. This approach involves expelling drugs directly from capsules at target sites, driven by various forces such as heat, electricity, gas, or magnetism. The capsule can release the drug continuously, intermittently, or at varying rates by sending signals that are controlled via a computer or preprogrammed for the target site in the GI tract.12 Delivery systems such as Enterion, Intellicap, SmartTab, and RoboCap are in research and development stages.
MCEs already are being used in clinical practice. Novel, magnetically controlled drug-delivering capsule endoscopes, developed by integrating a drug delivery system and an AI-assisted diagnostic system into an MCE, are designed to release active pharmaceutical ingredients into the GI tract.69 These capsules (31×11.6 mm, 3 g) consist of a 0.5-mL storage compartment, an electric pump for spraying, a drug delivery tube, a battery (10-hour life), and a camera, among other components.69 In porcine models, it was able to detect lesions using AI, maneuver to the target site via magnetic control, and orient precisely to aim the spray port.69
Tissue Sampling and Physiologic Sensing
Robotic capsule endoscopy systems perform tissue biopsies in animal models using mechanisms such as motorized blades, rotating razors, U-shaped clamps, and fine-needle devices.19 Robotic capsule endoscopy systems are being developed to address the shortcomings of conventional fluid and tissue collection methods via endoscopy, which can cause contamination or fail to accurately represent the microbiome of the entire GI tract.70 Smart capsules integrating various sensors to measure temperature, pH, pressure, intestinal gases, and hemoglobin are being studied.11,12,19 Such systems are in early stages of development and will need further validation for clinical adoption.
Role in Minimally Invasive Surgery
Advances in technology, particularly in power supply, could enable capsules to include endoscopic mucosal resection and endoscopic submucosal dissection functionalities, greatly expanding their clinical applications. A larger WCE robot could function as a carrier battleship and release clusters of nanorobot “soldiers” to perform surgery.11
References
- Richards DM, Irani M. Capsule endoscopy: past, present, and future. Gastroenterology & Endoscopy News. 2022;73(2):1-8.
- Adler SN. The history of time for capsule endoscopy. Ann Transl Med. 2017;5(9):194.
- Iddan G, Meron G, Glukhovsky A, et al. Wireless capsule endoscopy. Nature. 2000;405(6785):417.
- Tawheed A, Ismail A, Amer MS, et al. Capsule endoscopy: do we still need it after 24 years of clinical use? World J Gastroenterol. 2025;31(5):102692.
- Melson J, Trikudanathan G, Abu Dayyeh BK, et al. Video capsule endoscopy. Gastrointest Endosc. 2021;93(4):784-796.
- Lei II, Thorndal C, Manzoor MS, et al. The diagnostic accuracy of colon capsule endoscopy in inflammatory bowel disease—a systematic review and meta-analysis. Diagnostics (Basel). 2024;14(18):2056.
- Rosa B, Andrade P, Lopes S, et al. Pan-enteric capsule endoscopy: current applications and future perspectives. GE Port J Gastroenterol. 2023;31(2):89-100.
- Koulaouzidis A, Baatrup G. Current status of colon capsule endoscopy in clinical practice. Nat Rev Gastroenterol Hepatol. 2023;20(9):557-558.
- Spada C, Piccirelli S, Hassan C, et al. AI-assisted capsule endoscopy reading in suspected small bowel bleeding: a multicentre prospective study. Lancet Digit Health. 2024;6(5):e345-e353.
- Thorndal C, Selnes O, Lei II, et al. Retention of endoscopic capsules in diverticula: literature review of a capsule endoscopy rarity. Endosc Int Open. 2024;12(6):e788-E796.
- Cao Q, Deng R, Pan Y, et al. Robotic wireless capsule endoscopy: recent advances and upcoming technologies. Nat Commun. 2024;15(1):4597.
- Wei X, Xi P, Chen M, et al. Capsule robots for the monitoring, diagnosis, and treatment of intestinal diseases. Mater Today Bio. 2024;29:101294.
- Mehedi IM, Rao KP, Alotaibi FM, et al. Intelligent wireless capsule endoscopy for the diagnosis of gastrointestinal diseases. Diagnostics (Basel). 2023;13(8):1445.
- Jalayeri Nia G, Selnes O, Cortegoso Valdivia P, et al. An overview of emerging smart capsules using other-than-light technologies for colonic disease detection. Therap Adv Gastroenterol. 2024;17:17562848241255298.
- O’Hara FJ, Mc Namara D. Capsule endoscopy with artificial intelligence-assisted technology: real-world usage of a validated AI model for capsule image review. Endosc Int Open. 2023;11(10):E970-E975.
- Zhang Y, Zhang Y, Huang X. Development and application of magnetically controlled capsule endoscopy in detecting gastric lesions. Gastroenterol Res Pract. 2021;2021:2716559.
- Tal AO, Vermehren J, Albert JG. Colon capsule endoscopy: current status and future directions. World J Gastroenterol. 2014;20(44):16596-16602.
- Nakaji K. Colon capsule endoscopy: can it contribute to green endoscopy? World J Gastrointest Endosc. 2024;16(12):627-631.
- Rehan M, Al-Bahadly I, Thomas DG, et al. Smart capsules for sensing and sampling the gut: status, challenges and prospects. Gut. 2023;73(1):186-202.
- Al-Bayati K, Stone JK, Berzin TM. The use of artificial intelligence for endoscopic evaluation of the small bowel. Gastrointest Endosc Clin N Am. 2025;35(2):355-366.
- Parikh M, Tejaswi S, Girotra T, et al. Use of artificial intelligence in lower gastrointestinal and small bowel disorders: an update beyond polyp detection. J Clin Gastroenterol. 2025;59(2):121-128.
- Margalit Yehuda R, Davidov Y, Selinger L, et al. The visibility and performance of small bowel video capsule endoscopy with and without pre-procedural purge preparation in the same patients. J Gastroenterol Hepatol. 2025;40(6):1485-1491.
- Panenteric capsule endoscopy in gastrointestinal bleeding - time to change old habits? Curr Opin Gastroenterol. 2025;41(3):139-145.
- Velayos Jiménez B, Alcaide Suárez N, González Redondo G, et al. Impact of the endoscopist’s experience on the negative predictive value of capsule endoscopy. Gastroenterol Hepatol (English Edition). 2017;40(1):10-15.
- Riccioni ME, Urgesi R, Cianci R, et al. Negative capsule endoscopy in patients with obscure gastrointestinal bleeding reliable: recurrence of bleeding on long-term follow-up. World J Gastroenterol. 2013;19(28):4520-4525.
- Testoni SGG, Albertini Petroni G, Annunziata ML, et al. Artificial intelligence in inflammatory bowel disease endoscopy. Diagnostics (Basel). 2025;15(7):905.
- Tamilarasan AG, Tran Y, Paramsothy S, et al. The diagnostic yield of pan-enteric capsule endoscopy in inflammatory bowel disease: a systematic review and meta-analysis. J Gastroenterol Hepatol. 2022;37(12):2207-2216.
- Niv Y, Ilani S, Levi Z, et al. Validation of the Capsule Endoscopy Crohn’s Disease Activity Index (CECDAI or Niv score): a multicenter prospective study. Endoscopy. 2012;44(1):21-26.
- Yablecovitch D, Lahat A, Neuman S, et al. The Lewis score or the capsule endoscopy Crohn’s disease activity index: which one is better for the assessment of small bowel inflammation in established Crohn’s disease? Therap Adv Gastroenterol. 2018;11:1756283X17747780.
- Bourreille A, Jarry M, D’Halluin PN, et al. Wireless capsule endoscopy versus ileocolonoscopy for the diagnosis of postoperative recurrence of Crohn’s disease: a prospective study. Gut. 2006;55(7):978-983.
- Pons Beltran V, Nos P, Bastida G, et al. Evaluation of postsurgical recurrence in Crohn’s disease: a new indication for capsule endoscopy? Gastrointest Endosc. 2007;66(3):533-540.
- Hansen Lø, Thorndal C, Agache A, et al. Meckel’s diverticulum discovered by capsule endoscopy: a systematic review of case reports. Scand J Gastroenterol. 2025;60(5):414-420.
- Rossi RE, Poletti V, Masoni B, et al. The role of videocapsule endoscopy in the diagnosis and follow-up of celiac disease: a scoping review. Expert Rev Gastroenterol Hepatol. 2024;18(10):673-679.
- Pennazio M, Rondonotti E, Despott EJ, et al. Small-bowel capsule endoscopy and device-assisted enteroscopy for diagnosis and treatment of small-bowel disorders: European Society of Gastrointestinal Endoscopy (ESGE) Guideline - Update 2022. Endoscopy. 2023;55(1):58-95.
- Barret M, Malamut G, Rahmi G, et al. Diagnostic yield of capsule endoscopy in refractory celiac disease. Am J Gastroenterol. 2012;107(10):1546-1553.
- Atlas DS, Rubio-Tapia A, Van Dyke CT, et al. Capsule endoscopy in nonresponsive celiac disease. Gastrointest Endosc. 2011;74(6):1315-1322.
- Lacy BE, Mearin F, Chang L, et al. Bowel disorders. Gastroenterology. 2016;150(6):1393-1407.e1395.
- Huang L, Huang Z, Tai Y, et al. The small bowel diseases detected by capsule endoscopy in patients with chronic abdominal pain: a retrospective study. Medicine (Baltimore). 2018;97(8):e0025.
- Shim KN, Kim YS, Kim KJ, et al. Abdominal pain accompanied by weight loss may increase the diagnostic yield of capsule endoscopy: a Korean multicenter study. Scand J Gastroenterol. 2006;41(8):983-988.
- Sulbaran M, de Moura E, Bernardo W, et al. Overtube-assisted enteroscopy and capsule endoscopy for the diagnosis of small-bowel polyps and tumors: a systematic review and meta-analysis. Endosc Int Open. 2016;4(2):E151-163.
- Rasmussen MS, Hansen Lø, Deding U, et al. Applicability of colon capsule endoscopy for monitoring ulcerative colitis: a systematic review. Scand J Gastroenterol. 2025;60(4):336-342.
- Lei II, Arasaradnam R, Koulaouzidis A. Polyp matching in colon capsule endoscopy: pioneering CCE-colonoscopy integration towards an AI-driven future. J Clin Med. 2024;13(23):7034.
- Turvill J, Haritakis M, Pygall S, et al. Multicentre study of 10,369 symptomatic patients comparing the diagnostic accuracy of colon capsule endoscopy, colonoscopy and CT colonography. Aliment Pharmacol Ther. 2025;61(9):1532-1544.
- Baatrup G, Bjørsum-Meyer T, Kaalby L, et al. Choice of colon capsule or colonoscopy versus default colonoscopy in FIT positive patients in the Danish screening programme: a parallel group randomised controlled trial. Gut. 2025;74(10):1616-1623.
- Xiao ZG, Chen XQ, Zhang D, et al. Image detection method for multi-category lesions in wireless capsule endoscopy based on deep learning models. World J Gastroenterol. 2024;30(48):5111-5129.
- Rondonotti E, Pennazio M, Toth E, et al. How to read small bowel capsule endoscopy: a practical guide for everyday use. Endosc Int Open. 2020;8(10):E1220-E1224.
- Kim JE, Kim ER, Park JJ, et al. Comparative study of MiroCam MC2000 and PillCam SB3 in detecting small bowel bleeding: a multicenter prospective randomized crossover study. Gut Liver. 2025;19(4):569-578.
- Rezapour M, Amadi C, Gerson LB. Retention associated with video capsule endoscopy: systematic review and meta-analysis. Gastrointest Endosc. 2017;85(6):1157-1168.e2.
- Wang YC, Pan J, Liu YW, et al. Adverse events of video capsule endoscopy over the past two decades: a systematic review and proportion meta-analysis. BMC Gastroenterol. 2020;20(1):364.
- Cortegoso Valdivia P, Skonieczna-Zydecka K, Elosua A, et al. Indications, detection, completion and retention rates of capsule endoscopy in two decades of use: a systematic review and meta-analysis. Diagnostics (Basel). 2022;12(5):1105.
- Nemeth A, Wurm Johansson G, Nielsen J, et al. Capsule retention related to small bowel capsule endoscopy: a large European single-center 10-year clinical experience. United European Gastroenterol J. 2017;5(5):677-686.
- O’Hara F, McNamara D. Small-bowel capsule endoscopy-optimizing capsule endoscopy in clinical practice. Diagnostics (Basel). 2021;11(11):2139.
- Fernandez-Urien I, Carretero C, Gonzalez B, et al. Incidence, clinical outcomes, and therapeutic approaches of capsule endoscopy-related adverse events in a large study population. Rev Esp Enferm Dig. 2015;107(12):745-752.
- Liao Z, Gao R, Xu C, et al. Indications and detection, completion, and retention rates of small-bowel capsule endoscopy: a systematic review. Gastrointest Endosc. 2010;71(2):280-286.
- Bhattarai M, Bansal P, Khan Y. Longest duration of retention of video capsule: a case report and literature review. World J Gastrointest Endosc. 2013;5(7):352-355.
- Ling CR, Wang MJ, Zhuang W. Capsule retention for 7.5 years in Meckel’s diverticulum. Dig Endosc. 2017;29(3):386-387.
- Trasolini R, Byrne MF. Artificial intelligence and deep learning for small bowel capsule endoscopy. Dig Endosc. 2021;33(2):290-297.
- Cortegoso Valdivia P, Fantasia S, Kayali S, et al. Conventional small-bowel capsule endoscopy reading vs proprietary artificial intelligence auxiliary systems: systematic review and meta-analysis. Endosc Int Open. 2025;13:a25442863.
- Sidhu R, Shiha MG, Carretero C, et al. Performance measures for small-bowel endoscopy: a European Society of Gastrointestinal Endoscopy (ESGE) Quality Improvement Initiative – Update 2025. Endoscopy. 2025;57(4):366-389.
- Leenhardt R, Vasseur P, Li C, et al. A neural network algorithm for detection of GI angiectasia during small-bowel capsule endoscopy. Gastrointest Endosc. 2019;89(1):189-194.
- Aoki T, Yamada A, Aoyama K, et al. Clinical usefulness of a deep learning-based system as the first screening on small-bowel capsule endoscopy reading. Dig Endosc. 2020;32(4):585-591.
- Khan MA, Shafiq U, Hamza A, et al. A novel network-level fused deep learning architecture with shallow neural network classifier for gastrointestinal cancer classification from wireless capsule endoscopy images. BMC Med Inform Decis Mak. 2025;25(1):150.
- Dhali A, Kipkorir V, Maity R, et al. Artificial intelligence-assisted capsule endoscopy versus conventional capsule endoscopy for detection of small bowel lesions: a systematic review and meta-analysis. J Gastroenterol Hepatol. 2025;40(5):1105-1118.
- Saito H, Aoki T, Aoyama K, et al. Automatic detection and classification of protruding lesions in wireless capsule endoscopy images based on a deep convolutional neural network. Gastrointest Endosc. 2020;92(1):144-151.e1.
- Nadimi ES, Braun JM, Schelde-Olesen B, et al. Towards full integration of explainable artificial intelligence in colon capsule endoscopy’s pathway. Sci Rep. 2025;15(1):5960.
- Hansen Lø, Agache A, Koulaouzidis A. The future of minimally invasive GI and capsule diagnostics (REFLECT), October 2024. Diagnostics (Basel). 2025;15(7):859.
- Staib AH, Beermann D, Harder S, et al. Absorption differences of ciprofloxacin along the human gastrointestinal tract determined using a remote-control drug delivery device (HF-capsule). Am J Med. 1989;87(5A):66S-69S.
- Dhalla AK, Al-Shamsie Z, Beraki S, et al. A robotic pill for oral delivery of biotherapeutics: safety, tolerability, and performance in healthy subjects. Drug Deliv Transl Res. 2022;12(1):294-305.
- Wang W, Zhang K, Ming F, et al. A novel scheme for non-invasive drug delivery with a magnetically controlled drug delivering capsule endoscope. J Control Release. 2025;381:113591.
- Chen L, Gruzinskyte L, Jorgensen SL, et al. An ingestible self-polymerizing system for targeted sampling of gut microbiota and biomarkers. ACS Nano. 2020;14(9):12072-12081.
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.

Download to read this article in PDF document:![]() Capsule Endoscopy:: A Comprehensive Review of Current Applications and Future Directions
Capsule Endoscopy:: A Comprehensive Review of Current Applications and Future Directions