Director of Esophageal Subspecialty Group
Division of Gastroenterology and Hepatology
Mayo Clinic
Jacksonville, Florida
Barrett’s esophagus is a premalignant condition characterized by replacement of normal stratified squamous epithelium of the esophagus with specialized columnar-lined epithelium with goblet cells.1,2 BE is the only known precursor to esophageal adenocarcinoma and is thought to progress through a sequence of intestinal metaplasia to dysplasia to carcinoma. The prevalence of BE is estimated to be around 1% to 2% of the Western population.3
Current guidelines recommend upper endoscopy to screen for BE and EAC. Although there is some variability in BE screening guidelines, a common recommendation is to screen patients with chronic gastroesophageal reflux disease (GERD) and additional risk factors for BE, such as male sex, age older than 50 years, White race, central obesity, history of tobacco smoking, and a family history of BE or EAC in a first-degree relative.4,5 However, more recent guidelines recommend treating GERD as one of the risk factors and not a requirement for screening.6,7
Because the risk for progression to EAC varies significantly across histologic grades, patients with BE should undergo periodic surveillance every 3 to 5 years with a high-quality endoscopic exam and Seattle protocol biopsies using both high-definition white light endoscopy (HD-WLE) and chromoendoscopy. This approach has some limitations, such as low adherence to Seattle protocol biopsies and interobserver variability between pathologists in grading dysplasia. As a result, there is an emerging role for risk stratification tools to identify patients with BE who have a high risk for progression.
Ongoing management of BE primarily consists of endoscopic surveillance, with biopsies for patients who have a low risk of progressing to EAC and endoscopic eradication therapy (EET) for those at high risk for progression. EET consists of resection of any visible lesions, followed by ablation (radiofrequency ablation [RFA] or cryoablation) every 2 to 3 months until complete eradication of dysplasia and intestinal metaplasia.
This article presents a series of representative cases that illustrate the spectrum of management strategies for BE, highlighting best practices in diagnosis and management.
Case 1
A 59-year-old White man with a 20-year history of GERD presented to the clinic for evaluation. He described a 15-year history of heartburn and reflux symptoms that typically are worse after meals and when he is lying down. He takes over-the-counter antacids as needed to manage his symptoms. His body mass index (BMI) is 31 kg/m2; he has a 15 pack-year smoking history; and his family history includes a maternal uncle with esophageal cancer. The patient denied dysphagia, melena, and iron deficiency anemia.
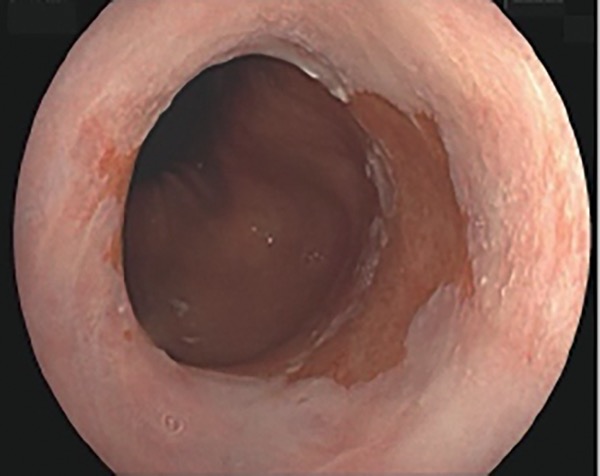
Upper endoscopy showed a 5-cm circumferential segment of salmon-colored mucosa above the gastroesophageal junction (GEJ), with tongues of salmon-colored mucosa extending proximally to 8 cm (Prague classification: C5M8) (Figure 1). No visible lesions were noted within the BE segment. Biopsies obtained using the Seattle protocol demonstrated specialized intestinal metaplasia without evidence of dysplasia, and a follow-up endoscopy in 3 years was recommended.
Case 1 Discussion
This case illustrates a patient with long-segment nondysplastic Barrett’s esophagus (NDBE) who underwent an upper endoscopy for screening due to multiple risk factors. His risk of progressing from NDBE to EAC is low, with an annual progression rate of 0.33% per year.8 However, because the risk is non-negligible in patients with long-segment (=3 cm) NDBE, endoscopic surveillance is recommended every 3 years4; a 5-year surveillance interval can be considered in patients with short-segment (<3 cm) NDBE.4
In addition, guidelines recommend that patients with BE receive maintenance therapy with a proton pump inhibitor at least once a day, regardless of GERD symptoms, for acid suppression and chemoprevention.4 Pooled observational data have shown that PPI therapy was associated with a 71% reduction in the risk for high-grade dysplasia (HGD) or EAC (adjusted odds ratio [OR], 0.29; 95% CI, 0.12-0.79).9
Use of histamine receptor antagonists do not confer the same benefit as PPI therapy in patients with BE. While the role for potassium competitive acid blockers in the BE population needs further evaluation, these agents can be considered as an alternative for patients who are refractory to or intolerant of PPI therapy.10
As in this case, patients must be informed about the nature of BE, the risks associated with progression to dysplasia or EAC based on the histologic grade of disease, and the available treatment options—including risks, benefits, and indication for PPI therapy—at the initial diagnosis. In addition, patients who are overweight should be counseled on weight loss, and smokers should be counseled on the importance of smoking cessation.
Case 2
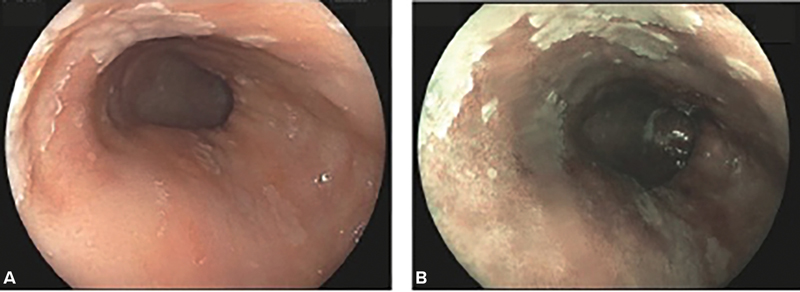
A 65-year-old woman with previously diagnosed short-segment BE (Prague classification: C0M2) presented for surveillance endoscopy. She reported a 10-year history of reflux symptoms and said she currently takes 40 mg of esomeprazole daily. She said her GERD symptoms are well controlled and that she very rarely has heartburn or reflux. Upper endoscopy showed a 2-cm segment of salmon-colored mucosa, with no evidence of esophagitis or visible lesions (Figure 2). Biopsies obtained per protocol were consistent with BE indefinite for dysplasia (IND), and a second GI pathologist agreed with the interpretation.
Case 2 Discussion
BE IND is found in about 8.4% of BE patients.11 Studies have shown variable progression rates to HGD/EAC in patients with IND.11 Some risk factors for progression are longer-segment BE, multifocal areas of IND, and persistent IND on multiple exams. However, many patients show no dysplasia on follow-up exams, especially after being treated with high-dose PPI therapy.
Sometimes this lack of finding is due to regression of dysplasia. Alternatively, the lack of an IND finding on a subsequent exam could be a result of interobserver variation among pathologists in distinguishing dysplasia from reactive changes. On the initial diagnosis of IND, it is important to confirm the diagnosis by expert pathology review. Patients should be placed on high-dose, twice-a-day PPI therapy, with repeat high-quality exam by an expert endoscopist within 6 months to rule out any prevalent HGD/ EAC.4 Those with IND should undergo a surveillance exam every year until there is a change in dysplasia grade.10 If histology regresses to NDBE on a surveillance exam, then follow-up surveillance for NDBE every 3 to 5 years, depending on BE segment length, is appropriate.
A meticulous and standardized approach to endoscopic evaluation of BE is essential for accurate diagnosis and surveillance.12 Identification of key esophageal landmarks, such as the diaphragmatic hiatus, GEJ, and squamocolumnar junction, is critical for orientation and future exams. The use of a distal attachment cap, especially in patients with known dysplasia, improves visualization, while thorough mucosal cleaning with a water jet and careful suctioning removes debris and reduces trauma. Controlled insufflation and desufflation can help reveal subtle mucosal abnormalities.
Adequate inspection time of the BE segment (1 minute per centimeter of BE) and visualization of the gastric cardia in retroflexion are crucial for detecting dysplasia. HD-WLE and chromoendoscopy enhance visualization of mucosal and vascular patterns. The Prague classification should be used to describe the BE segment and the Paris classification to describe any visible lesions. In addition, biopsies should be obtained per Seattle protocol from any targeted areas, as well as every 2 cm in patients without dysplasia and every 1 cm in those with a history of dysplasia, with a partially deflated esophagus.12
Case 3
A 73-year-old man with a 6-year history of BE presented for routine surveillance. He reported a history of chronic GERD, for which he has been taking 40 mg of pantoprazole twice a day, with good symptom control. His comorbidities include cirrhosis complicated by ascites, coronary artery disease with prior stent placement, stage III chronic kidney disease, and peripheral vascular disease. The patient has a BMI of 41 and lives independently with some mobility limitations. He denied dysphagia, weight loss, hematemesis, and melena.
An EGD showed a 6-cm segment of BE (Prague classification: C4M6). Biopsies taken per Seattle protocol demonstrated low-grade dysplasia (LGD), with diagnosis confirmed by an expert GI pathologist. The diagnosis of LGD was discussed with the patient, including next steps in treatment of LGD with EET, as well as the risks and benefits of this approach.
The patient expressed concern about undergoing therapeutic endoscopies for EET, the potential for adverse events given his comorbidities, and logistical challenges with travel and post-procedure care. Due to these concerns, he chose to undergo surveillance, with the understanding that he would reconsider treatment options if his condition progressed.
Case 3 Discussion
Current guidelines of the American Gastroenterological Association suggest that EET should be used rather than surveillance in patients with confirmed BE with LGD.10 However, surveillance is a reasonable management strategy in patients for whom concern about risks associated with the procedure outweigh the potential benefits. When discussing management with EET versus surveillance for LGD, shared decision-making is critical.
Annual rates of progression in patients with LGD have been reported to be 5.7%.13 Although pooled data from 3 randomized controlled trials did not show a statistically significant decrease in progression to EAC in LGD patients who underwent EET versus surveillance,14-16 observational studies have suggested a statistically significant decrease in EAC with EET.17,18 Cost-effectiveness analyses also support EET for confirmed LGD.19
However, 28% to 66% of LGD will regress to NDBE on subsequent exams with optimal acid suppression.20,21 This could be due to sampling error or interobserver variability in the histologic interpretation of LGD, but either is an argument for surveillance over EET.
Patients with LGD who choose surveillance should maintain high-dose acid-suppressive therapy and undergo high-quality exams by an expert endoscopist at 6-month intervals for 1 year and annually thereafter. For LGD patients who choose EET, the primary goal is complete eradication of intestinal metaplasia (CEIM), as for HGD. If complete eradication of dysplasia is achieved after a few EET sessions or there is recurrence of nondysplastic BE after CEIM, the residual nondysplastic disease can be reasonably managed with surveillance rather than ongoing EET.
Case 4
A 56-year-old man with a known long-segment BE, previously under surveillance at a different center, presented with a 3-month history of intermittent solid-food dysphagia. He reported a long-standing history of GERD and said he was on daily omeprazole but admitted to poor medication adherence. The patient had not undergone endoscopic surveillance for 5 years due to relocation.
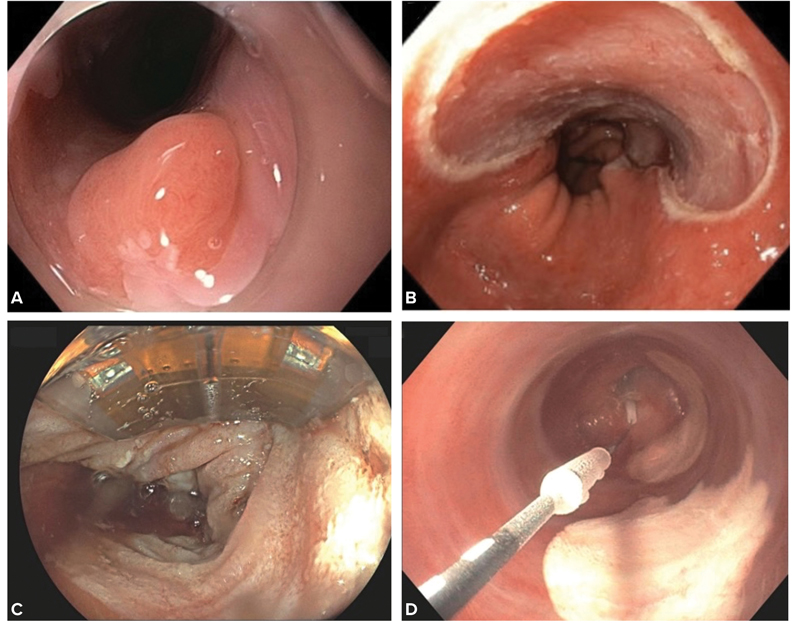
Because the patient’s symptoms were poorly controlled, PPI dosing was increased to twice daily. Surveillance EGD revealed a 5-cm BE segment (Prague classification: C3M5) with a 15-mm area of nodularity at 39 cm from the incisors (Figure 3A). Endoscopic mucosal resection (EMR) was performed (Figure 3B). Histopathology showed a focus of stage T1a EAC in a background of HGD. The deep margin of the resection specimen was free of dysplasia and carcinoma. A CT scan of the chest/abdomen/pelvis showed no evidence of distant disease or lymphadenopathy. The patient underwent sequential sessions of ablative therapy with RFA until CEIM (Figure 3C).
Case 4 Discussion
For patients with HGD, EET is recommended because rates of progression to HGD/EAC have been reported to be as high as 19%.16 EET with RFA or cryoablation also is recommended in patients with stage T1a EAC.4,10
RFA has a larger body of efficacy and safety data and remains the first-line ablative modality for EET.15,16 Cryoablation is considered for salvage therapy for patients who are refractory to RFA or those with EAC due to the greater depth of treatment penetration achieved with this ablative technique.22 The 2 available cryoablation devices are liquid nitrogen spray cryotherapy using the truFreeze system (Steris) and the C2 CryoBalloon (Pentax) (Figure 3D). EMR or endoscopic submucosal dissection (ESD) can be used for endoscopic resection of visible lesions in EET.
Although the majority of visible lesions in BE can be managed with EMR, ESD has utility in treating large, bulky neoplastic lesions or those with features suspicious for deeper invasion. EET typically is performed in sessions that occur every 2 to 3 months until CEIM. It is important that patients are placed on high-dose acid-suppressive therapy before initiation of EET because most cases refractory to EET are due to inadequate acid suppression.10
After CEIM, patients should enter a post-CEIM surveillance pathway, with periodic endoscopies with biopsies to monitor for disease recurrence at intervals dictated by the baseline severity of dysplasia. In patients with baseline HGD or intramucosal carcinoma, surveillance is recommended at 3, 6, and 12 months after CEIM and annually thereafter.4 For patients with LGD, endoscopic surveillance is recommended at 1 year after CEIM and every 2 years thereafter.
In post-CEIM surveillance exams, biopsies should be obtained from the GEJ and distal esophagus (2-5 cm proximal to the GEJ) and placed in separate jars. Intestinal metaplasia of the cardia is not considered to be clinically significant and does not require treatment.
References
- Barrett NR. Chronic peptic ulcer of the oesophagus and ‘oesophagitis.’ Br J Surg. 1950;38(150):175-182.
- Que J, Garman KS, Souza RF, et al. Pathogenesis and cells of origin of Barrett’s esophagus. Gastroenterology. 2019;157(2):349-364.e1.
- Qumseya BJ, Bukannan A, Gendy S, Ahemd Y, Sultan S, Bain P, et al. Systematic review and meta-analysis of prevalence and risk factors for Barrett’s esophagus. Gastrointest Endosc. 2019;90(5):707-17.e1.
- Shaheen NJ, Falk GW, Iyer PG, et al. Diagnosis and management of Barrett’s esophagus: an updated ACG guideline. Am J Gastroenterol. 2022;117(4):559-587.
- Fitzgerald RC, di Pietro M, Ragunath K, et al. British Society of Gastroenterology guidelines on the diagnosis and management of Barrett’s oesophagus. Gut. 2014;63(1):7-42.
- Muthusamy VR, Wani S, Gyawali CP, et al. AGA clinical practice update on new technology and innovation for surveillance and screening in Barrett’s esophagus: expert review. Clin Gastroenterol Hepatol. 2022;20(12):2696-2706.e1.
- Qumseya B, Sultan S, Bain P, et al. ASGE guideline on screening and surveillance of Barrett’s esophagus. Gastrointest Endosc. 2019;90(3):335-359.e2.
- Desai TK, Krishnan K, Samala N, et al. The incidence of oesophageal adenocarcinoma in non-dysplastic Barrett’s oesophagus: a meta-analysis. Gut. 2012;61(7):970-976.
- Singh S, Garg SK, Singh PP, et al. Acid-suppressive medications and risk of oesophageal adenocarcinoma in patients with Barrett’s oesophagus: a systematic review and meta-analysis. Gut. 2014;63(8):1229-1237.
- Rubenstein JH, Sawas T, Wani S, et al. AGA clinical practice guideline on endoscopic eradication therapy of Barrett’s esophagus and related neoplasia. Gastroenterology. 2024;166(6):1020-1055.
- Krishnamoorthi R, Mohan BP, Jayaraj M, et al. Risk of progression in Barrett’s esophagus indefinite for dysplasia: a systematic review and meta-analysis. Gastrointest Endosc. 2020;91(1):3-10.e3.
- Kolb JM, Wani S. Endoscopic eradication therapy for Barrett’s oesophagus: state of the art. Curr Opin Gastroenterol. 2020;36(4):351-358.
- Singh S, Manickam P, Amin AV, et al. Incidence of esophageal adenocarcinoma in Barrett’s esophagus with low-grade dysplasia: a systematic review and meta-analysis. Gastrointest Endosc. 2014;79(6):897-909.e4; quiz 83.e1, 83.e3.
- Barret M, Pioche M, Terris B, et al. Endoscopic radiofrequency ablation or surveillance in patients with Barrett’s oesophagus with confirmed low-grade dysplasia: a multicentre randomised trial. Gut. 2021;70(6):1014-1022.
- Phoa KN, van Vilsteren FG, Weusten BL, et al. Radiofrequency ablation vs endoscopic surveillance for patients with Barrett esophagus and low-grade dysplasia: a randomized clinical trial. JAMA. 2014;311(12):1209-1217.
- Shaheen NJ, Sharma P, Overholt BF, et al. Radiofrequency ablation in Barrett’s esophagus with dysplasia. N Engl J Med. 2009;360(22):2277-2288.
- Small AJ, Araujo JL, Leggett CL, et al. Radiofrequency ablation is associated with decreased neoplastic progression in patients with Barrett’s esophagus and confirmed low-grade dysplasia. Gastroenterology. 2015;149(3):567-e14.
- Wolf WA, Pasricha S, Cotton C, et al. Incidence of esophageal adenocarcinoma and causes of mortality after radiofrequency ablation of Barrett’s esophagus. Gastroenterology. 2015;149(7):1752-1761.e1.
- Omidvari AH, Ali A, Hazelton WD, et al. Optimizing management of patients with Barrett’s esophagus and low-grade or no dysplasia based on comparative modeling. Clin Gastroenterol Hepatol. 2020;18(9):1961-1969.
- Sharma P, Falk GW, Weston AP, et al. Dysplasia and cancer in a large multicenter cohort of patients with Barrett’s esophagus. Clin Gastroenterol Hepatol. 2006;4(5):566-572.
- Thota PN, Lee HJ, Goldblum JR, et al. Risk stratification of patients with Barrett’s esophagus and low-grade dysplasia or indefinite for dysplasia. Clin Gastroenterol Hepatol. 2015;13(3):459-465.e1.
- Sengupta N, Ketwaroo GA, Bak DM, et al. Salvage cryotherapy after failed radiofrequency ablation for Barrett’s esophagus-related dysplasia is safe and effective. Gastrointest Endosc. 2015;82(3):443-448.`
Copyright © 2025 McMahon Publishing, 545 West 45th Street, New York, NY 10036. Printed in the USA. All rights reserved, including the right of reproduction, in whole or in part, in any form.
Download to read this article in PDF document:![]() Diagnosis and Management of Barrett’s Esophagus: A Case-Based Approach
Diagnosis and Management of Barrett’s Esophagus: A Case-Based Approach