SAN DIEGO—Treating younger patients with inflammatory bowel disease can be particularly challenging, with considerations such as off-label treatments, concerns about how treatment will affect growth, and the need for shared decision-making that includes patients as well as their parents.
Athos Bousvaros, MD, MPH, a professor of pediatrics at Harvard Medical School and the associate director of the IBD Center at Boston Children’s Hospital, gave a presentation on how he approaches difficult pediatric cases at DDW 2025.
The first step? “Think about what you’re doing and why you’re doing it,” Dr. Bousvaros said, keeping two overarching goals of treatment in mind: improving or normalizing quality of life and preventing future disease complications such as abscess or stricture, cancer, and growth failure. Measures such as C-reactive protein, calprotectin and endoscopic healing “are just things to get you there.”
To develop a successful treatment algorithm for IBD in pediatric patients, it’s important to keep several key concepts in mind, Dr. Bousvaros said.
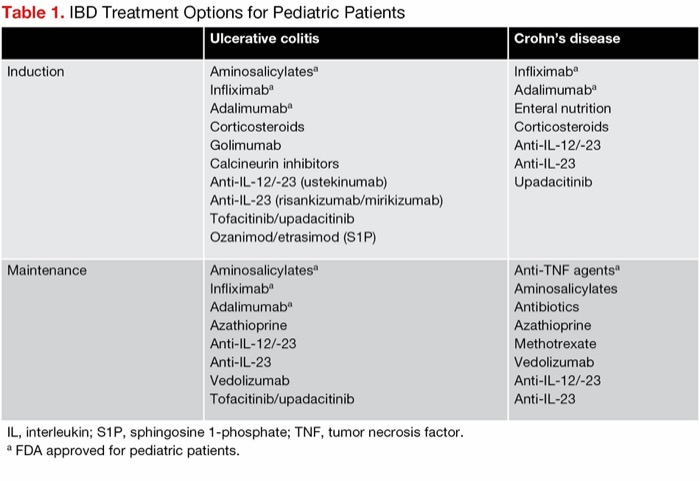
| Table 1. IBD Treatment Options for Pediatric Patients | ||
| Ulcerative colitis | Crohn’s disease | |
|---|---|---|
| Induction |
|
|
| Maintenance |
|
|
| IL, interleukin; S1P, sphingosine 1-phosphate; TNF, tumor necrosis factor. a FDA approved for pediatric patients. | ||
Off-Label Use Required
Because most IBD treatments are not approved for younger patients, often, off-label use of medication is medically necessary (Table 1). Dr. Bousvaros shared a consensus statement from the American Academy of Pediatrics that emphasized the term “off-label” does not imply an “improper, illegal, contraindicated or investigational use,” as the purpose of off-label use is first and foremost to benefit the patient.
“This is really important because insurance companies often try to deny off-label use as being ‘experimental.’ It’s not experimental,” he said. However, the downsides to off-label use is that there are no “FDA-level data,” so safety and efficacy are not as well established. In addition, using drugs off label instead of enrolling patients in clinical trials may delay regulatory approval.
“Given the ethical issues intrinsic to trials involving children, including concerns about giving a sick child a placebo and exposing a child who cannot provide informed consent to an investigational drug, FDA approval for children often trails well behind approval of the same medication in adults,” said Erica Brenner, MD, MSCR, a pediatric gastroenterologist and an assistant professor at UNC Children’s Hospital, in Chapel Hill, N.C. “A new drug may not yet be FDA approved in children even though it is safe and effective.”
To put these considerations into a practical context, Dr. Bousvaros described a hypothetical case of a hospitalized patient with ulcerative colitis who had not responded to anti–tumor necrosis factor therapy or steroids. Often, he said, the first resort is to increase the dose of the anti-TNF agent. If that fails, calcineurin inhibitors or Janus kinase (JAK) inhibitors are an option for sick inpatients (those with bloody diarrhea, severe pain or anemia), whereas those who are less sick may receive anti-interleukin (IL)-23 therapies or vedolizumab (Entyvio, Takeda), which have more favorable safety profiles but work more slowly, he said.
Another treatment option for pediatric UC patients is tacrolimus, Dr. Bousvaros said, citing a study that found it was comparable to infliximab for preventing surgery (J Pediatr Gastroenterol Nutr 2023;77[2]:222-227). Data on JAK inhibitors are limited, but he cited a study in adults that showed upadacitinib (Rinvoq, AbbVie) was an effective treatment after infliximab (Am J Gastroenterol 2024 Mar 27. doi:10.14309/ajg.0000000000002674). Another study, also in adults, found tofacitinib (Xeljanz, Pfizer) was effective as first-line therapy, although it is only FDA approved as second-line therapy (Am J Gastroenterol 2024;119[7]:1365-1372), and a study of tofacitinib in pediatric patients refractory to biologic agents showed good eight- and 24-week outcomes (Inflamm Bowel Dis 2025;31[2]:425-431), indicating that these agents eventually may be options to use in pediatric settings to prevent escalation to anti-TNF therapy, he added.
In addition, preliminary pediatric data on mirikizumab (Omvoh, Lilly) are strong, Dr. Bousvaros said, with 38.5% of pediatric patients with UC achieving remission, a higher rate than was seen in adults (Gastroenterol Hepatol [NY] 2023;19[7 suppl 3]:8-9).
2 Drugs May Be Better Than 1
Dr. Bousvaros highlighted data from the VEGA trial, which compared the anti-TNF golimumab (Simponi, Johnson & Johnson) alone, the anti-IL-23 guselkumab (Tremfya, Johnson & Johnson) and a combination of the two in adults with UC. At week 28, about 21% of the golimumab group, 31% of the guselkumab group and 48% of the combination group achieved remission (Lancet Gastroenterol Hepatol 2023;8[4]:307-320).
“These patients are not your classic sick inpatients—they tend to be a little bit more well—but combos work,” he said.
Returning to the hypothetical UC patient, Dr. Bousvaros offered an algorithm:
- Optimize anti-TNF therapy through therapeutic drug monitoring.
- If they get better, keep them on the anti-TNF agent.
- If they’re really sick, consider a calcineurin or JAK inhibitor or combination therapy.
- If they’re less sick, consider an IL-23, vedolizumab, a sphingosine-1-phosphate, a JAK inhibitor or combination therapy.
Regarding combinations, Dr. Brenner noted that data “remain severely limited for dual advanced therapy in pediatric IBD, but case series are emerging that suggest they may be effective for select children with refractory disease [Dig Dis Sci 2024;69(5):1826-1833].”
How to Approach CD
Crohn’s disease refractory to anti-TNF therapy requires a different approach, Dr. Bousvaros said, emphasizing the importance of careful review of patient data. In colonic CD, it’s particularly important to confirm that the patient has CD instead of UC.
“I’ve had so many patients sent to me where the diagnosis is probably UC, and it was based on some weak data,” he said. Once CD is confirmed, it’s important to identify the phenotype to guide therapy. For example, he said, inflammatory disease may be treated with enteral nutrition, medication or surgery, whereas strictures and perforations necessitate dilation or surgery. Perianal disease may require antibiotics, medication (typically biologics) and, sometimes, surgery.
“Keep in mind a child only has one opportunity to grow, and that’s puberty,” Dr. Bousvaros said. “Get aggressive in puberty.”
Dr. Brenner agreed. “Active inflammation blocks growth and can delay puberty,” she noted. “Once a patient’s growth plates have fused, they lose the opportunity to achieve catch-up growth. Therefore, I see the teenage years as a particularly critical time to keep a patient’s IBD in deep remission.”
Several medication options exist for a young patient with CD refractory to anti-TNF therapy, Dr. Bousvaros said, so when weighting treatment options for such a patient, it’s important to keep a few things in mind (Table 2).
| Table 2. Key Considerations When Treating Kids With Anti-TNF Refractory CD | |
| Medication | Considerations |
|---|---|
| Vedolizumab |
|
| Anti-IL-12/-23 and anti-IL-23 agents (ustekinumab, risankizumab/mirikizumab) |
|
| Upadacitinib |
|
| EEN, exclusive enteral nutrition; EIMs, extra-intestinal manifestations; IL, interleukin; TNF, tumor necrosis factor. | |
He cited VEDOkids, a large European study that found comparable or superior efficacy to that seen in adults, although remission rates in UC were higher than in CD (Lancet Gastroenterol Hepatol 2023;8[1]:31-42). Ustekinumab (Stelara, Johnson & Johnson) also has been studied in pediatric patients and has shown good efficacy; however, Dr. Bousvaros emphasized that data from these studies should be interpreted with caution, as dropouts lead to survivorship bias. A small, single-center study of upadacitinib in patients between 12 and 17 years of age revealed a strong six-month steroid-free remission rate in CD, Dr. Bousvaros said, although data demonstrated it is more effective in UC (Inflamm Bowel Dis 2024;30[11]:2057-2063).
Keep Surgery on the Table
With the array of treatment options for IBD, Dr. Bousvaros said surgery is too often overlooked as an indispensable part of IBD treatment. Citing a 1990 study published before the age of IBD medications, he said resection of limited disease led to an increase in growth rate from 2 cm per year to 8 cm per year (Br J Surg 1990;77[8]:891-894).
“Surgery works,” he said. “Keep in mind that the main cause of growth failure are three things: inflammation, prolonged steroid use and malnutrition. All of these can be helped with surgery.”
Colonic disease is more difficult. If a patient or parent does not want a colectomy, consider diversion, which can help height velocity and weight gain and reduce hospitalization, but it’s a “careful and difficult decision,” Dr. Bousvaros said.
Connotations and fears related to surgery can be challenging to navigate, he added, but patients often underestimate how much better they will feel. “The most common question I’ve had after the surgery from a patient is why didn’t I do this earlier,” he added.
Practice Shared Decision-Making
Optimal treatment begins with shared decision-making, which should address FDA approval, efficacy, safety, cost and convenience.
“Patients are more often worried about medication side effects than disease control,” Dr. Bousvaros said, particularly in milder cases. While most IBD medications generally are safe, he said, “risk tolerance varies,” and patients’ views may be based on anecdotes rather than data. In addition, he said, conversations are essential to ensure that patients and their families understand the importance of disease control and providers understand contextual details, such as insurance coverage and travel plans, that should inform treatment decisions.
“Often there is no single one right drug,” he said. “It’s more a matter of what the factors are that help you make the decision.”
—Katie Prince
Dr. Bousvaros reported financial relationships with AbbVie, Arena/Pfizer, Bristol Myers Squibb, Janssen, Lilly and Takeda.
This article is from the October 2025 print issue.