Massachusetts General Hospital Assistant Professor of Anesthesia
Harvard Medical School Boston
I remember the discovery of glucagon-like peptide-1 (GLP-1) sometime in the late 1980s. It came as the answer to a finding so strange, so blatantly dissonant, that even a junior medical student like myself, who was devouring science journals at the American Library in Bucharest, Romania, was finding it deeply disturbing: During glucose tolerance tests, the same amount of glucose triggered a higher insulin release if ingested, as opposed to directly injected into the vein. For anyone who understood the concept of bioavailability, this was simply impossible. The process, we thought at that time, was simple: The beta cells sense blood glucose and release insulin; the greater the insulin release, the greater the triggering hyperglycemia must have been. How can an oral load bring more sugar into the blood than putting that sugar directly there? It was impossible. They called it “the incretin effect.”1
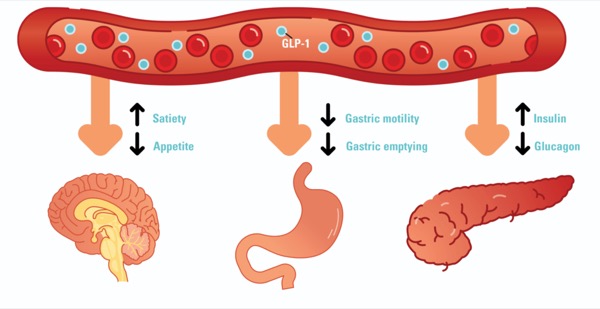
What the framework was missing was understanding the role of incretins, hormones that are released from the intestinal mucosa and modulate insulin secretion. GLP-1 is such a hormone, and is responsible for up to 60% of insulin release.2 It took a long time—and a beautiful story of medical discovery, industry and perseverance—to develop this hormone into a drug, which was not only safer than exogenous insulin for patients with diabetes, but, at higher doses, it reduced appetite and caused weight loss. During an obesity epidemic, this was a holy grail for pharmacologists. And it all started with an observation that initially did not make sense.
What also did not make any sense for me, one day at the end of September last year, was to see a young man anesthetized in front of me and having a stomach full of half-digested food3; he had fasted for 18 hours. This was supposed to be his fifth uncomplicated upper endoscopy in less than two years. He had told us of no digestive discomfort. And yet, here we were. A murky fluid was pouring from his mouth; he was coughing, aspirating, desaturating. Sandy, a superbly knowledgeable, experienced and talented CRNA, and I knew exactly what to do. More propofol, a syringe of succinylcholine, rapid sequence intubation, recruitment, bronchoscopy, meticulously clean all the food remains from the bronchi. We knew that the following hours would be critical, and we observed him in the ICU. Nothing happened. He was fine. He was lucky.
Two months prior, he started taking semaglutide for weight loss. Like most of us at that time, I regarded these drugs as safe for anesthesia. But GLP-1 is known to inhibit gastric emptying, so maybe it was because of this, I thought. The Canadian Journal of Anesthesia was the third journal I submitted the case report to, and this time it was received by an editor—dare I say a visionary—called Philip Jones, MD, who saw its potential and, critically, assigned it to one of the most remarkable peer reviewers I have ever encountered.3 I will never know their name. They shared my concern, told me of old studies that I was not aware of and painstakingly questioned each of my initial risk-mitigating ideas. I had the chance to discuss my very first thoughts with someone who was knowledgeable, logical, open and grounded, and who, anonymously, became instrumental to the early development of this story. If you are reading this, thank you.
One year later, four more case reports,4-7 two retrospective studies8,9 and one prospective study10 confirmed my initial concerns. By the time you read these words, there may be more. Our own retrospective study at Massachusetts General Hospital is in press, in the Canadian Journal of Anesthesia. In 90 endoscopy patients on GLP-1 agonists, we saw a six-time increase in the incidence of a full stomach after overnight fasting compared with controls. This is essentially the same as that previously reported by Silveira et al,9 on a smaller scale. In our patients, the increased risk seemed independent of the GLP-1 agonist dose and duration of treatment, and led to several intraprocedural emergent intubations, but these preliminary conclusions need to be tested in larger cohorts.
So, “full stomach.” With advanced notice, this is completely manageable. We anesthetize patients with full stomachs all the time: in emergencies, parturients and patients with bowel obstruction. “Either awake or rapid sequence” I tell my residents. “No deep MAC [monitored anesthesia care], no LMAs [laryngeal mask airways].”
Or perhaps we can avoid this altogether if we hold the drug perioperatively. However, the optimal holding duration is unclear. For semaglutide, an American Society of Anesthesiologists Task Force has recommended one week. For a drug taken once weekly, I cannot see how this will make any difference. We are essentially telling patients to take the drug as usual, but instead of taking the next dose just come to have the operation. The patients of Silveira et al9 held semaglutide for 10 days and still had a high incidence of full stomachs. My initial proposal3 was for a hold of 3.3 half-lives, 24 days, and I remain to this opinion. If this is too onerous for patients, we can bridge with liraglutide, a once-daily agonist, and hold that for two days before surgery. But I fully accept that these are only theoretical calculations for the time necessary to achieve a 90% reduction in drug blood levels, and we have no positive evidence that this is the period of time necessary and sufficient to bring gastric emptying to normal. We should investigate this directly, perhaps by using gastric ultrasound to assess the stomach contents. This technique could also be used for risk stratification, if more studies would confirm that, in adequately trained hands, it has a reassuring sensitivity.
We need to perform this research rapidly and rigorously and continue the scientific momentum achieved in the last months. And most importantly, we must make no mistakes. You see, that late 1980s moment was in fact the second time that incretins were discovered. Their existence was first postulated in the 1930s, but the first experiments were flawed, and when those initial errors was demonstrated by Andrew Ivy, MD, a foremost endocrinologist and discoverer of cholecystokinin, the entire idea came into disrepute.1 No one would even touch on the topic for the next 25 years—25 years lost. A generation. Sometimes mistakes are costly.
This is why I would rather act, as Dr. Jones says, with an abundance of caution.11 We do not want to be wrong. Let us advise on a longer holding period for now and relax the precautionary measures later if new scientific evidence indicates it. And let us cherish the uncertainty we feel right now; it is uncomfortable, I know, but it is also invaluable because this is what drives research. And to our patients, I would disclose it fully. I would tell them the concerns we have, offer them the information available at this point, what we think but do not know for sure yet and involve them in the clinical decision of how to proceed. Tell them that we are in a transition period right now and it will take some time to get our feet fully underneath us. They will understand.
Dr. Hobai reported no relevant financial disclosures.
References
- Rehfeld .F. The origin and understanding of the incretin concept. Front Endocrinol (Lausanne). 2018;9:387.
- Kreymann B, Williams G, Ghatei MA, et al. Glucagon-like peptide-1 7-36: a physiological incretin in man. Lancet. 1987;2(8571):1300-1304.
- Klein SR, Hobai IA. Semaglutide, delayed gastric emptying, and intraoperative pulmonary aspiration: a case report. Can J Anaesth. 2023;70(8):1394-1396.
- Gulak MA, Murphy P. Regurgitation under anesthesia in a fasted patient prescribed semaglutide for weight loss: a case report. Can J Anaesth. 2023;70(8):1397-1400.
- Weber M, Siddarthan I, Mack PF. Clinically significant emesis in a patient taking a long-acting GLP-1 receptor agonist for weight loss. Br J Anaesth. 2023;131(2):e37-e39.
- Beam WB, Hunter Guevara LR. Are serious anesthesia risks of semaglutide and other GLP-1 agonists under recognized? Case reports of retained solid gastric contents in patients undergoing anesthesia. APSF Newsletter. 2023;38(3).
- Wilson PR, Bridges KH, Wilson SH. Particulate gastric contents in patients prescribed glucagon-like peptide 1 receptor agonists after appropriate perioperative fasting: a report of 2 cases. A A Pract. 2023;17(8):e01712.
- Kobori T, Onishi Y, Yoshida Y, et al. Association of glucagon-like peptide-1 receptor agonist treatment with gastric residue in an esophagogastroduodenoscopy. J Diabetes Investig. 2023;14(6):767-773.
- Silveira SQ, da Silva LM, de Campos Vieira Abib A, et al. Relationship between perioperative semaglutide use and residual gastric content: a retrospective analysis of patients undergoing elective upper endoscopy. J Clin Anesth. 2023;87:111091.
- Sherwin M, Hamburger J, Katz D, et al. Influence of semaglutide use on the presence of residual gastric solids on gastric ultrasound: a prospective observational study in volunteers without obesity recently started on semaglutide. Can J Anaesth. 2023;70(8):1300-1306.
- Jones PM, Hobai IA, Murphy PM. Anesthesia and glucagon-like peptide-1 receptor agonists: proceed with caution! Can J Anaesth. 2023;70(8):1281-1286.
This article is from the December 2023 print issue.