The increased risk for metachronous colorectal neoplasms after polypectomy in patients with metabolic dysfunction–associated steatotic liver disease can be ameliorated in a dose-dependent manner if metabolic dysfunction is improved, according to the results of a large study from Taiwan.
“Interventions for reducing metabolic dysfunction during the surveillance interval can be regarded as a strategy of primary prevention against metachronous [colorectal] neoplasms in MASLD patients,” said investigator Wei-Yuan Chang, MD, of National Taiwan University Hospital, Taipei. He noted that “a minor reduction [in metabolic dysfunction] was associated with a 23% decreased risk, while a major reduction was associated with a 37% decreased risk.”
Importance of Assessing Metabolic Status
“After polypectomy, surveillance interval is stratified by the individual’s risk for a metachronous lesion, but what is the impact of an individual’s metabolic status on this risk?” Dr. Chang asked. “We know the five-year prevalence of advanced metachronous lesions ranged from 8% to 37% in individuals with different index colonoscopy findings. However, for patients with metabolic dysfunction, we think such risk could be increased. Careful surveillance of this population is recommended.”
He added that a second reason for assessing metabolic status is for primary prevention, assuming improving metabolic dysfunction can reduce risk. For example, weight loss can ameliorate risk for advanced colorectal neoplasms in people with central obesity.
In their retrospective study, reported at DDW 2024 (abstract 398), Dr. Chang and his co-investigators stratified the risk for metachronous colorectal neoplasms in people with MASLD and tested whether an improvement in metabolic dysfunction might prevent these lesions. The population included average-risk people 40 years of age and older who underwent screening or surveillance colonoscopy between 2009 and 2021. Baseline metabolic profile was obtained at screening colonoscopy, while metabolic status at follow-up was obtained on the day of surveillance colonoscopy.
Hepatic steatosis was diagnosed by ultrasound on the day of screening or surveillance colonoscopy. MASLD was diagnosed in patients with hepatic steatosis and at least one cardiometabolic risk factor (overweight or central obesity, impaired fasting glucose, hypertension, hypertriglyceridemia, or low serum high-density lipoprotein). The baseline severity of metabolic dysfunction was assessed according to the number of risk factors observed on the day of screening. Improvement in dysfunction was defined as a reduction in the number of factors at the time of surveillance.
Overall, 2,333 people diagnosed with MASLD also had at least one adenoma on screening colonoscopy. Metabolic dysfunction was considered mild (one to two risk factors) for 760 participants, moderate (three factors) for 758 and severe (four to five factors) for 815.
During a mean follow-up of four years, 539 individuals demonstrated mild improvement in metabolic status (a reduction in one risk factor); 215 moderately improved (a reduction in at least two risk factors); and the remaining 1,579 achieved no metabolic improvement.
Presence of Metachronous Neoplasms
Of the 2,331 patients, 1,131 (48.5%) had one or more metachronous neoplasms and 129 (5.4%) had advanced neoplasms. Those with any metachronous neoplasm were somewhat older (median, 58 vs. 56 years; P<0.01) and more likely to have a family history of colorectal cancer (20% vs. 16%; P=0.01).
In the multivariate analysis, each one-item increment in cardiometabolic risk factors was associated with a 6% increase in metachronous colorectal neoplasms. Risk also was increased by 24% in patients with a family history of CRC, by 29% in males and by 3% for each one-year increase in age.
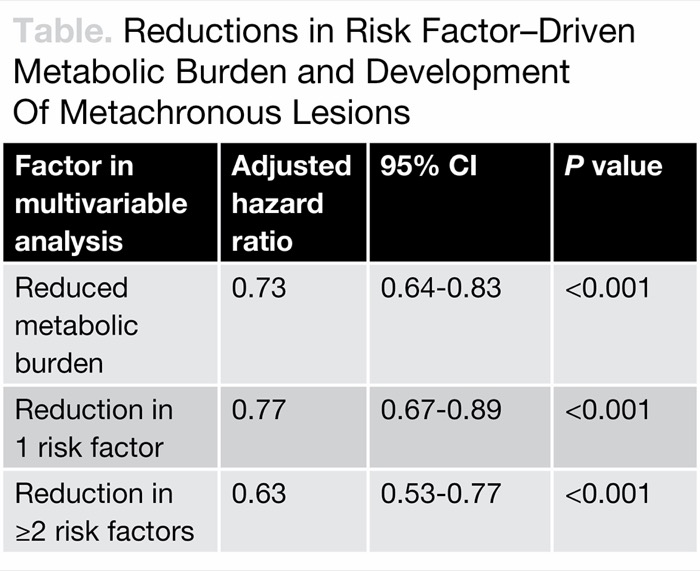
In those whose metabolic burden was reduced during the surveillance interval, the risk for a metachronous lesion was reduced by 27% (P<0.001). The dose–response magnitude of these risk factor reductions was reflected in varying degrees of risk reduction (Table). The type of intervention leading to the reduction in metabolic burden was not determined.
| Table. Reductions in Risk Factor–Driven Metabolic Burden and Development Of Metachronous Lesions | |||
| Factor in multivariable analysis | Adjusted hazard ratio | 95% CI | P value |
|---|---|---|---|
| Reduced metabolic burden | 0.73 | 0.64-0.83 | <0.001 |
| Reduction in 1 risk factor | 0.77 | 0.67-0.89 | <0.001 |
| Reduction in =2 risk factors | 0.63 | 0.53-0.77 | <0.001 |
The investigators also found that “for patients with major reductions of their uncontrolled metabolic burden, risk for advanced metachronous lesions could also be significantly reduced by 55%” (hazard ratio, 0.45; 95% CI, 0.24-0.82; P=0.01), Dr. Chang noted. Some risk factors—primarily hypertension and hyperlipidemia—carried more predictive weight than others, he said.
The researchers noted that future goals are to develop prediction models based on metabolic dysfunction components and other factors associated with risk for metachronous colorectal neoplasms.
Commenting on the findings after the presentation, Elena Martinez, PhD, the Sam W. Walton Endowed Chair for Cancer Research at the University of California, San Diego, said her research group published a study 15 years ago that examined body mass index and metachronous colorectal neoplasms (Am J Epidemiol 2009;169[6]:657-666). “Back then, our study was not as sophisticated as yours today, in terms of our metabolic definitions, but it was prospective and our findings were similar,” she said.
Commenting on the study in an interview, Jonathan Stine, MD, MSc, the Fatty Liver Program director at Penn State Health, said, “This important study reveals a strong link between MASLD and increased risk of colorectal neoplasms after polypectomy, emphasizing the need for aggressive metabolic management to reduce this risk.”
Weight loss and cardiometabolic control could be worthy interventions, Dr. Stine said, suggesting that the integration of metabolic optimization into CRC prevention protocols may significantly improve outcomes. “Future research should focus on developing predictive models that incorporate metabolic parameters to better stratify risk and tailor surveillance strategies for these high-risk patients,” Dr. Stine added. “This would ideally include some evaluation of body composition, given the complex relationship between adiposity, MASLD and oncologic risk.”
Drs. Chang, Martinez and Stine reported no relevant financial disclosures.
This article is from the December 2024 print issue.