Section Editors
University of Washington School of Medicine
Seattle, Washington
Virginia Tech Carilion School of Medicine
Roanoke, Virginia
Universidad de La República
Montevideo, Uruguay
Contributors
Carilion Memorial Hospital
Virginia Tech Carilion School of Medicine
Roanoke, Virginia
Virginia Tech Carilion School of Medicine
Roanoke, Virginia
Universidad de La República
Montevideo, Uruguay
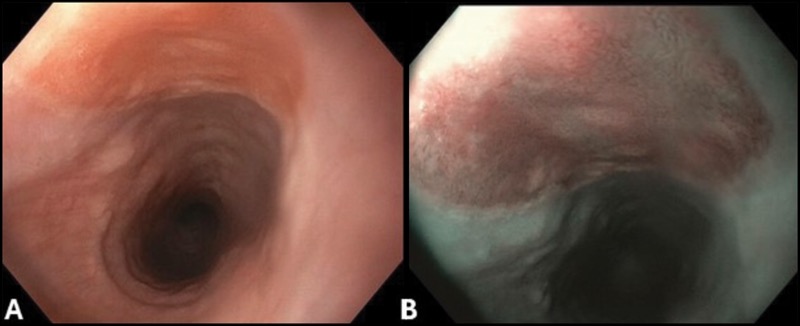
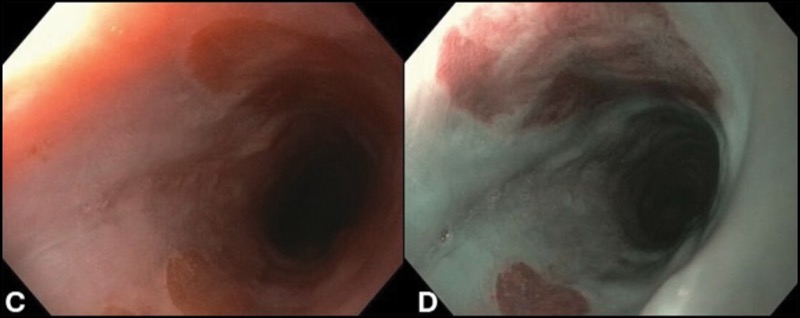
A 38-year-old woman presented with a one-year history of globus sensation, intermittent dysphagia, and cough. An esophagogastroduodenoscopy showed 2 areas of stomach heterotopia, or “inlet patches,” in the upper esophagus (Figure, panels A-D).
Because this patient was symptomatic, a decision was made to ablate the inlet patches. Multiple studies have shown good results for argon plasma coagulation (APC) in the ablation of symptomatic inlet patches.
It is important to carefully characterize these lesions endoscopically, and re-ablation biopsies are recommended to evaluate the type of stomach tissue because research has described cases of adenocarcinoma arising in inlet patches. Since inlet patches are common, and since there is no definitive evidence that they contribute to nonspecific cough and dysphagia, this kind of endoscopic therapy should typically only be considered if there is strong evidence of a causal relationship.
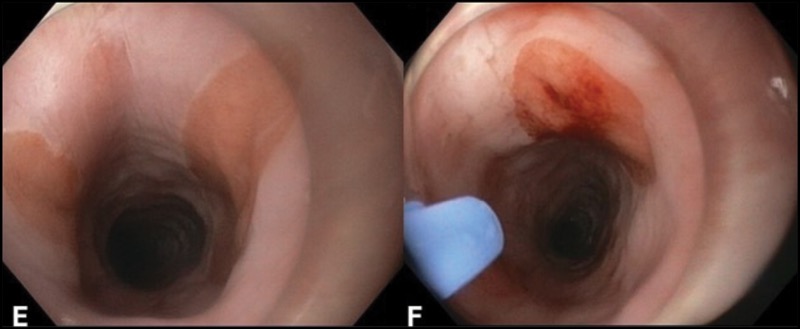
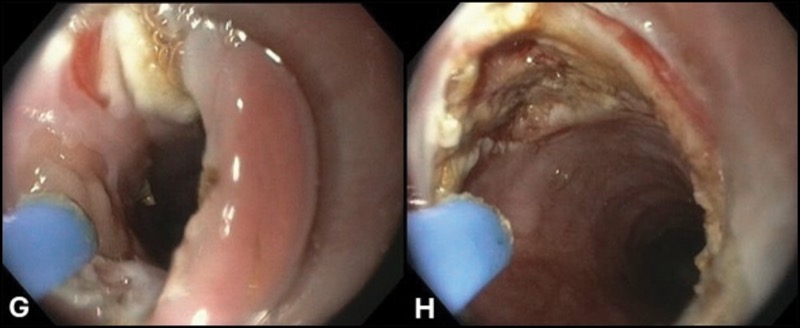
Although some experts ablate the lesions using the argon plasma catheter through the scope, we prefer to use a distant transparent cap (Figure, panels E-G).
Inlet patches are usually located very close to the upper esophageal sphincter. Thus, a cap will keep the esophageal entrance open, while allowing targeting of the inlet patches with the tip of the argon plasma catheter (Figure, panels F and G).
In addition, the cap allows for scraping off of ablated tissue and applying a second or third round of APC. The aim of APC is to change the color of the mucosa to a brown color (Figure, panel H), not to carbonize or char it black, because this may result in deep-tissue injury and subsequent esophageal stenosis.
The settings used are 40 to 50 watts with a 0.8-L flow. However, one can apply lower wattage and flow if the endoscopist can achieve a controlled mucosal ablation with the movements of the scope and catheter.
After the procedure, the patient was placed on viscous lidocaine as needed and proton pump inhibitors twice a day. She was restricted to a liquid diet for 6 hours, followed by a soft diet for 1 day. She resumed a regular diet 48 hours after the procedure.
Dr. Mergener is a member of the Gastroenterology & Endoscopy News editorial board.
See endocollab.com for more information, including videos, quick tips, and lectures on these and other endoscopy techniques.
This article is from the February 2025 print issue.


