SAN DIEGO—The safety of simethicone during colonoscopy has been questioned in recent years, but a study from a large Department of Veterans Affairs medical center presented at DDW 2025 showed that simethicone can improve efficiency when delivered via water reservoir.
“We found significant benefit in efficiency with simethicone usage in the endoscope’s water reservoir, with no significant increase in infection rate,” said Michael Perrin, MD, a fourth-year resident at Baylor College of Medicine, in Houston, who conducted the research with senior investigator Scott Larson, MD, PhD, an assistant professor of medicine at Baylor and a gastroenterologist at the Michael E. DeBakey VA Medical Center. Compared with simethicone delivered via a biopsy port/channel, reservoir-delivered simethicone was associated with significant reductions in intubation time, withdrawal time and sedation use, even after adjusting for covariates, Dr. Perrin reported at the meeting (abstract 250).
Potential Risk for Infection?
Simethicone, a defoaming agent often used to improve mucosal visualization during the colonoscopy, works as a surfactant, reducing the surface tension of the bubbles and causing them to burst. In recent years, concerns have been raised about using simethicone because the silicone in simethicone is not dissolved by water or detergents, and simethicone residue may remain in the endoscope channels despite high-level disinfection, leading to retained moisture that can harbor bacteria.
“While no infection was directly linked to the phenomenon, there is a theoretical risk,” Dr. Perrin said. These concerns “put the spotlight on simethicone as a possible contributing factor in endoscopy-related bacterial infections.”
Simethicone typically is delivered in one of two ways: injected via syringe as a bolus through the endoscope’s biopsy channel or injected on demand from the water bottle reservoir. The latter can be cleaned manually with a brush prior to automated endoscope reprocessing, Dr. Perrin noted. Concerns about the potential for infection triggered the issuance of a Jan. 1, 2024, national VA guidance document prohibiting simethicone in the water bottle reservoir and mandating that simethicone be given in aliquots via the biopsy port.
This guidance was the catalyst for the study reported at DDW, Dr. Perrin said. “No study has ever shown actual increased infection risk with reservoir simethicone use. The aim of our study was to determine whether its use in reservoirs is associated with post-procedure infection and whether this change impacted procedure time, sedation usage and adenoma detection rate [ADR],” he explained.
The investigators conducted a retrospective cohort study of all patients undergoing colonoscopy for any indication at Houston’s Michael E. DeBakey VA Medical Center during September 2023 (using the reservoir system) and April 2024 (using the biopsy channel system). In September 2023, simethicone was routinely added to the water reservoir. In April 2024, after the new guidance, 10-mL aliquots were given through the biopsy channel on request. With the method of simethicone administration as the independent variable, the investigators compared average withdrawal and cecal intubation times as primary outcomes. Secondary outcomes were ADR, post-procedure 30-day infection rate and sedation use.
Of 446 total colonoscopies, 211 (47.3%) used reservoir simethicone and 235 (52.7%) used channel simethicone. There were no significant differences between these groups with respect to demographic or procedural factors.
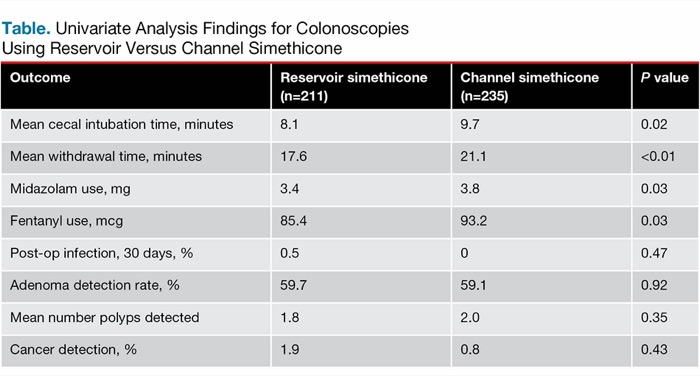
| Table. Univariate Analysis Findings for Colonoscopies Using Reservoir Versus Channel Simethicone | |||
| Outcome | Reservoir simethicone (n=211) | Channel simethicone (n=235) | P value |
|---|---|---|---|
| Mean cecal intubation time, minutes | 8.1 | 9.7 | 0.02 |
| Mean withdrawal time, minutes | 17.6 | 21.1 | <0.01 |
| Midazolam use, mg | 3.4 | 3.8 | 0.03 |
| Fentanyl use, mcg | 85.4 | 93.2 | 0.03 |
| Post-op infection, 30 days, % | 0.5 | 0 | 0.47 |
| Adenoma detection rate, % | 59.7 | 59.1 | 0.92 |
| Mean number polyps detected | 1.8 | 2.0 | 0.35 |
| Cancer detection, % | 1.9 | 0.8 | 0.43 |
Reservoir Outcomes Superior
Mean cecal intubation and withdrawal times were significantly longer with the channel approach than with the reservoir approach (9.7 vs 8.1 minutes and 21.1 vs. 17.6 minutes, respectively) (Table). In addition, use of midazolam and use of fentanyl were higher with the channel approach. There were no significant differences in ADR, cancer detection rates or postoperative infection.
“Although there was no significant difference in ADR, the time taken to visualize polyps was significantly higher in the channel group, as reflected by the longer intubation and withdrawal times, suggesting an increased efficiency in the reservoir group,” Dr. Perrin added.
In the multivariate analysis, the researchers adjusted for extensive biopsy (at least two in more than two colon segments), endoscopy performed by a gastroenterology fellow, patient age, terminal ileum intubation and number of polyps removed. They found that channel simethicone was a significant predictor of increased intubation time (regression coefficient, 1.57; P=0.03), increased withdrawal time (2.86; P<0.01) and sedation usage (1.95; P<0.01).
Potential Real-World Significance Huge
Dr. Perrin explained the surprising “real-world” clinical significance of a 3.5-minute increase in withdrawal time per procedure using the channel approach. “In a scenario in which we used reservoir simethicone, given its 3.5-minute shorter withdrawal time, for an endoscopist performing 12 colonoscopies per day, the time savings would be 42 minutes per day, or about one colonoscopy.
“That’s enough time to complete another scope,” he noted. “This is 144 additional colonoscopies per year if the endoscopist performs procedures three days per week and has four weeks of vacation.”
Dr. Perrin discussed several important considerations related to the use of simethicone that will require further investigation. “Given the risk simethicone poses when scopes are inadequately sterilized, we believe it’s important to maintain strict adherence to best practices for high-level disinfection. This includes the use of a forced air drying system, which has proven effective in removing retained water in endoscopes if done properly.” In addition, Dr. Perrin said the use of pre-procedure versus intra-procedure simethicone or a combination of the two should be studied, noting that the Colon Endoscopy Bubble Scale (CEBuS) can be used to standardize evaluation of bubbles.
Findings Open the Door For Further Study
Mazen Elatrache, MD, who moderated the session during which the results were presented, told Gastroenterology & Endoscopy News that the study presents “newer data” that follow up on findings from 10 years ago when infection risk was attributed to the use of simethicone in colonoscopy.
Given the retrospective nature of the study, Dr. Elatrache said that he does not consider the findings to be sufficient for drawing conclusions about safety. However, he said, this data “should open the door for further studies to clearly define if there is an actual increased risk of infection with reservoir simethicone use or not.”
Dr. Elatrache, an advanced therapeutic endoscopist at Henry Ford Health, in Detroit, added that the study suggests procedural efficiency is improved “when simethicone is delivered via the water reservoir. Today, clinical care is so much about optimization, about the endoscopy unit running efficiently, and about being time- and cost-saving. We focus on start times and end times and room turnover times. If larger and randomized studies in the future can demonstrate the lack of correlation between infections and reservoir simethicone use and also show improved efficacy,” he said, “then perhaps we’ll need to reconsider this.”
But for now, the latest multisociety guidance on HLD and sterilization recommends against putting simethicone in the irrigation water bottle (see story above).
—Caroline Helwick
Drs. Elatrache and Perrin reported no relevant financial disclosures.
This article is from the September 2025 print issue.