PuraStat gel reduced the risk for delayed bleeding after endoscopic mucosal resection and endoscopic submucosal dissection to treat large colonic polyps, by 88% and 72%, respectively, a recent study found. In patients at highest risk, the intervention also saved money.
“The prophylactic use of PuraStat [3-D Matrix] is safe, and it definitely reduces the delayed bleeding rate from what is expected,” said Pradeep Bhandari, MD, a professor of gastroenterology at Portsmouth Hospitals University NHS Trust, in England, who reported the findings at DDW 2025 (abstract 220).
Delayed bleeding, the most common complication after endoscopic resection of large proximal colonic polyps (=20 mm), occurs in almost 10% of patients, Dr. Bhandari said, but clipping reduces the bleeding rate to about 3% (Gastroenterology 2019;157[4]:977-984.e3).
PuraStat, a self-assembling synthetic hemostatic viscous solution that polymerizes and forms bonds that create a “scaffold” atop the surface of the bleeding vessel, can be used to treat intraprocedural bleeding or can be used as prophylactic therapy to prevent delayed postprocedural gastrointestinal bleeding.
In the current study, the researchers evaluated the clinical impact and cost-effectiveness of the hemostatic product in preventing delayed bleeding after EMR and ESD of large colonic polyps. Delayed bleeding was defined as bleeding after 24 hours and within 28 days of the index endoscopic procedure that could be directly attributed to the procedure and results in hospital admission/medical intervention, including endoscopic treatment.
Post-Procedure Prophylactic Administration
The prospective cohort study was conducted at eight tertiary referral centers on 293 patients who were undergoing endoscopic resection of polyps larger than 20 mm in the right colon or, for patients receiving antithrombotic agents, in any location. PuraStat was applied prophylactically at the conclusion of EMR and ESD. The delayed bleeding rates in the prospective cohort were compared with the theoretical expected delayed bleeding risk for the cohort, using validated delayed bleeding risk scores: GSEED-RE2 for EMR and the LIMOGES for ESD. Cost-effectiveness of the prophylactic strategy was determined and compared with the treatment of bleeding when it occurs without prophylaxis.
The EMR group included 191 patients with a mean polyp size of 44 mm; 22% of patients were receiving antithrombotic agents. Most polyps were in the colon (72%), while 28% were in the rectum; high-grade dysplasia or submucosal dysplasia was noted in 27% of cases.
The ESD group included 102 patients with a mean polyp size of 48 mm, and 15% were on antithrombotic agents. In this cohort, most polyps were in the rectum (59%) and 41% were in the colon. High-grade dysplasia or submucosal invasion was noted in 42% of cases.
Effect and Cost-Effectiveness
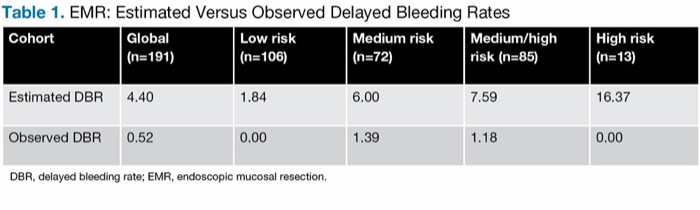
Based on the GSEED-RE2 prediction score, the estimated delayed bleeding rate in the EMR control group, in which no prophylactic hemostatic agent was applied, was 4.4% for the whole cohort and 7.6% in the medium- to high-risk subset. The observed rate of delayed bleeding in the PuraStat group, in contrast, was 0.5% in the whole cohort, a reduction of 88% (Table 1).
| Table 1. EMR: Estimated Versus Observed Delayed Bleeding Rates | |||||
| Cohort | Global (n=191) | Low risk (n=106) | Medium risk (n=72) | Medium/high risk (n=85) | High risk (n=13) |
|---|---|---|---|---|---|
| Estimated DBR | 4.40 | 1.84 | 6.00 | 7.59 | 16.37 |
| Observed DBR | 0.52 | 0.00 | 1.39 | 1.18 | 0.00 |
| DBR, delayed bleeding rate; EMR, endoscopic mucosal resection. | |||||
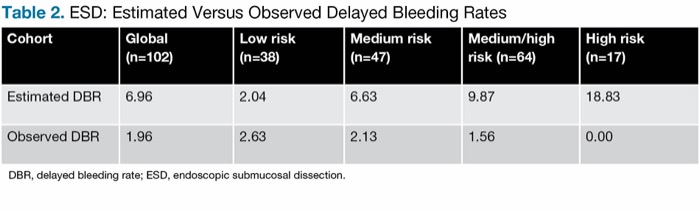
For the ESD group, the LIMOGES score was used to calculate the estimated delayed bleeding rates, which were reduced by 72% with PuraStat (Table 2).
| Table 2. ESD: Estimated Versus Observed Delayed Bleeding Rates | |||||
| Cohort | Global (n=102) | Low risk (n=38) | Medium risk (n=47) | Medium/high risk (n=64) | High risk (n=17) |
|---|---|---|---|---|---|
| Estimated DBR | 6.96 | 2.04 | 6.63 | 9.87 | 18.83 |
| Observed DBR | 1.96 | 2.63 | 2.13 | 1.56 | 0.00 |
| DBR, delayed bleeding rate; ESD, endoscopic submucosal dissection. | |||||
Dr. Bhandari noted that PuraStat is an expensive product; therefore, the researchers aimed to ascertain which patients would most benefit from its use. The study showed that while it is clinically effective at all levels of risk, it is cost-effective only in high-risk patients, although in medium- to high-risk patients, its benefit probably outweighs its slight additional cost, Dr. Bhandari maintained.
After analyzing the economics of its use—including the “real” cost of delayed bleeding in his center’s cohort after each procedure in all risk groups, the actual cost of the product in all risk groups, and the estimated cost of delayed bleeding without prophylaxis—the researchers arrived at the economic impact of this intervention per patient.
In high-risk patients undergoing EMR, they calculated a cost savings of 420 pounds ($558) per patient to reduce delayed bleeding from 16.37% (estimated) to 0.01% (observed). In high-risk patients undergoing ESD, the savings would be 468 pounds ($621) per patient to reduce bleeding risk from 18.83% (estimated) to 0.01% (observed).
In patients at medium- to high-risk, while no money would be actually saved, bleeding would be notably diminished for the small additional cost. An extra 57 pounds ($76) per patient would reduce bleeding from 7.59% (estimated) to 1.18% (observed) in the EMR cohort, and an additional 30 pounds ($40) would reduce bleeding from 9.87% (estimated) to 1.56% (observed) in the ESD cohort.
“The use of PuraStat in the high-risk group for both EMR and ESD will certainly save money, but there are very few patients in this group, so the practical approach is to use it in medium- to high-risk patients, where there will be a very small excess cost but a significant reduction in delayed bleeding,” Dr. Bhandari said.
Mixed Results
Heiko Pohl, MD, a professor of medicine at Dartmouth Hitchcock Medical Center, in Lebanon, New Hampshire, pointed out that the recent PURPLE trial was stopped for futility after no benefit was shown for PuraStat in preventing bleeding after EMR (Gut 2025;74[7]:1103-1111).
Dr. Bhandari noted that he and his co-investigators had raised several methodological issues about the PURPLE study in a letter they submitted to Gut (2025;74[8]:1351-1352). He said that these issues “limit the interpretability and clinical applicability of its findings” For one thing, he said, clipping and coagulation were allowed on patients, rendering PuraStat more of a “rescue strategy” than prophylaxis. “And the trial was prematurely halted following an interim analysis involving only 200 patients. … This pragmatic interim analysis might have been conducted too early on too small a sample size, which could potentially underpower the study to detect a clinically relevant effect.”
In another letter in the same issue, Albeniz et al also posed questions regarding the sample size and raised the possibility that the PURPLE study may have been underpowered, he added (Gut 2025;74[8]:1352-1354).
Dr. Pohl told Gastroenterology & Endoscopy News that the use of prediction models for bleeding has its strengths and weaknesses. “But even without a prediction model, the absolute proportion of patients who bled in this study was extremely low—lower than you’d expect in the right colon, which would be 3% to 5% with clipping and 10% without clipping. Here, their bleeding rate was less than 2%. This could be for a number of reasons, and one of those could be the use of PuraStat. But in some other case series, the numbers are not as promising.”
Dr. Bhandari said he believes the encouraging results were largely because his team understands how to use the product. “PuraStat is so easy to use that physicians often don’t pay attention to details,” he explained. “Lots of little things can make a difference.”
—Caroline Helwick
Dr. Bhandari reported financial relationships with 3-D Matrix, Boston Scientific, Fujifilm, NEC (Europe), Olympus, and Pentax. Dr. Pohl reported financial relationships with Olympus and Pentax.
This article is from the November 2025 print issue.