WASHINGTON—Computer-assisted detection of colorectal polyps, based on the application of artificial intelligence, has rapidly gained traction and is being used in clinical practice. In multiple sessions at DDW 2024, speakers discussed the many ways in which AI has, and has not, enhanced colonoscopy.
Rapid Evolution
“The volume and breadth of information about AI in endoscopy is astounding,” said Vani J.A. Konda, MD, the medical director of the Baylor Scott & White Center for Esophageal Diseases, in Dallas. For instance, four different versions of GI Genius (Medtronic) have been marketed. The computer-assisted detection (CADe) program for colonoscopy was first based on 3 million images, but later versions trained on 9 million, then 13 million and finally “double that,” she said. And more programs have entered the market, such as CAD EYE (Fujifilm), DISCOVERY (Pentax) and ENDO-AID (Olympus).
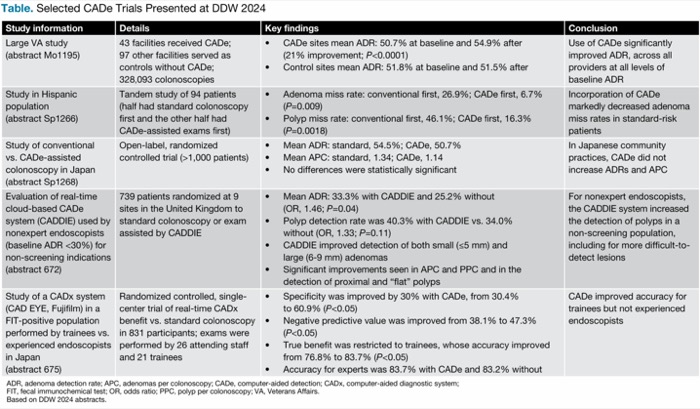
Similarly, literature citations pertaining to AI-assisted endoscopy numbered around 200 in 2019 but approached 1,200 in 2023, she noted. A group led by Cesare Hassan, MD, PhD, an associate professor of gastroenterology at Humanitas University, in Milan, who has published extensively on the use of AI in colonoscopy, first reported a meta-analysis of six randomized controlled trials, but their most recent meta-analyses in 2023 included 21 trials. “The field is moving so fast, we need a ‘living’ meta-analysis,” Dr. Hassan commented. Multiple studies were presented at the DDW meeting (Table).
| Table. Selected CADe Trials Presented at DDW 2024 | |||
| Study information | Details | Key findings | Conclusion |
|---|---|---|---|
| Large VA study (abstract Mo1195) | 43 facilities received CADe; 97 other facilities served as controls without CADe; 328,093 colonoscopies |
| Use of CADe significantly improved ADR, across all providers at all levels of baseline ADR |
| Study in Hispanic population (abstract Sp1266) | Tandem study of 94 patients (half had standard colonoscopy first and the other half had CADe-assisted exams first) |
| Incorporation of CADe markedly decreased adenoma miss rates in standard-risk patients |
| Study of conventional vs. CADe-assisted colonoscopy in Japan (abstract Sp1268) | Open-label, randomized controlled trial (>1,000 patients) |
| In Japanese community practices, CADe did not increase ADRs and APC |
| Evaluation of real-time cloud-based CADe system (CADDIE) used by nonexpert endoscopists (baseline ADR <30%) for non-screening indications (abstract 672) | 739 patients randomized at 9 sites in the United Kingdom to standard colonoscopy or exam assisted by CADDIE |
| For nonexpert endoscopists, the CADDIE system increased the detection of polyps in a non-screening population, including for more difficult-to-detect lesions |
| Study of a CADx system (CAD EYE, Fujifilm) in a FIT-positive population performed by trainees vs. experienced endoscopists in Japan (abstract 675) | Randomized controlled, single-center trial of real-time CADx benefit vs. standard colonoscopy in 831 participants; exams were performed by 26 attending staff and 21 trainees |
| CADe improved accuracy for trainees but not experienced endoscopists |
| ADR, adenoma detection rate; APC, adenomas per colonoscopy; Based on DDW 2024 abstracts. | |||
Strong Evidence From Randomized Trials
Randomized controlled trials have consistently shown that the use of CADe decreases the rate of missed adenomas and increases the adenoma detection rate (ADR) and adenomas per colonoscopy. A meta-analysis by Dr. Hassan and his team involving 21 randomized trials and more than 18,000 patients showed that AI assistance reduced adenoma miss rates from 35% to 16%, a relative risk reduction of 55% (risk ratio, 0.45; 95% CI, 0.35-0.58) (Ann Intern Med 2023;176[9]:1209-1220). The ADR was 44% with CADe and 36% with standard exams, a relative benefit of 24% with AI assistance (95% CI, 1.16-1.33).
Real-World Practice Results Not as Good
The positive benefits of AI in non-randomized trials, however, are mixed, with many showing no improvement in the detection of adenomas with AI assistance. “More than 20 randomized controlled trials have consistently demonstrated these improvements, but in non-randomized trials the improvements have not been consistent,” said Michael B. Wallace, MD, MPH, the Fred C. Andersen Professor of Medicine at Mayo Clinic in Florida, Jacksonville.
He and other speakers noted that non-randomized studies are likely to be hampered by variations in patient populations, bowel prep quality, endoscopist expertise, work environments and time constraints as well as to suffer from recording and reporting biases.
An often-cited study comparing community endoscopists performing colonoscopy with and without the use of AI showed no increase in ADR over baseline (Gastroenterology 2023;164[3]:481-483). Lead author Uri Ladabaum, MD, a professor of medicine and the director of the Gastrointestinal Cancer Prevention Program at Stanford University School of Medicine, in California, suggested that the problem is one of poor mucosal exposure. “We think it wasn’t so much that endoscopists were missing polyps on the screen but [rather that] they were just not doing a great exam. They were not getting good mucosal exposure,” he said.
This possibility “raises the broader question of the risk of overreliance on these technologies,” Dr. Wallace said. “AI only works if the lesion is on the screen. … For the most part, it is finding what we’ve already exposed. I think we need correlative technologies that tell us to slow down, to look behind the folds.”
Use of one such device (Endocuff Vision, Olympus), used along with CADe, boosted ADRs in recent studies, from 52% to 59% (Gastrointest Endosc 2023;97[3]:507-516) and from 44% to 50% (Gastroenterology 2023;165[1]:244-251).
Optimal Targets for AI
Speakers agreed that the best targets for AI assistance are the low detectors, that is, endoscopists with ADRs on the lower side, who are missing polyps that can lead to interval cancers. Dr. Hassan noted that in the NordICC trial (N Engl J Med 2022;387[17]:1547-1556), most of the interval cancers occurred in patients seen by endoscopists with low ADRs. “The impression is that low detectors get the most benefit from AI,” he said, “although low detectors may just be bad at navigation.”
Can AI also improve a trainee’s performance? The results of studies are mixed. “Trainees may be improving their performance, but they may not be learning to do colonoscopies,” Dr. Hassan maintained. “Performance is not learning. Stop the AI and determine if the trainee has developed the necessary skills.”
Shortcomings of AI Assistance
Current versions are good at detecting adenomas in general, but not in detecting advanced adenomas in particular. Potential reasons could be that advanced lesions are more visible (i.e., are larger) and that earlier AI versions evaluated in those trials were not adequately trained, Dr. Hassan suggested.
“What we are really finding are small polyps, 3 to 5 mm. There’s a slight decrease in the miss rate of larger ones (>10 mm), but the benefit is not statistically significant” in most studies, Dr. Wallace said.
“We’ve not been able to tease out whether an increase in ADR that is solely based on finding more 5-mm polyps is really beneficial,” Dr. Hassan added.
CADe also is associated with a lower miss rate for non-polypoid lesions, Dr. Wallace said. “But not all flat lesions are the same. The 3- to 4-mm sessile polyp is likely of very low clinical importance. It’s the 10- to 20-mm sessile serrated lesions, especially in the right colon, that could progress rapidly to cancer,” he stressed. “We’ve not been able to provide an improvement in the detection of advanced, large, lateral spreading polyps, although you could argue that any improvement in ADR reduces the risk of colorectal cancer.”
The less prevalent lesion types also may be missed more often because the AI model was not adequately trained to detect them, or because some studies were not powered sufficiently to detect significant differences in finding these more uncommon types, noted Jonathan Cohen, MD, a clinical professor of medicine at NYU Grossman School of Medicine, in New York City.
Finally, a common complaint among clinicians are false alerts, which when compounded across an exam can be annoying and fatiguing. “At the end of day, you may wish the system hadn’t found all those 1- to 2-mm polyps,” Dr. Wallace said. “They are likely not relevant, but when there’s a green box around them you feel obliged to remove them.”
Set Goals, Be Realistic
Dr. Konda suggested that practices should set goals for implementing AI with an understanding of its limitations. “It does not make up for a bad bowel prep or not seeing behind the folds. You can’t create different contexts or conclusions in situations outside of what the system was designed for,” Dr. Konda said. “AI also does not tell you what to do, … and it’s an adjunct, not a competitor. Ultimately, the physician is responsible.” With this in mind, endoscopists should ask: What gap needs to be addressed? What cases are ideal for its use, and when will we use it? How will we track the impact of AI?
AI Applications Will Greatly Expand
Although there are many provisos surrounding about the current role of AI in endoscopy, experts expect it to someday offer many additional benefits: histologic diagnosis and grading; risk stratification; ascertainment of depth of lesions; assistance in resection of lesions; documentation of the procedure’s completeness; achievement of adequate photo documentation; and evaluation of the quality of the procedure.
Beyond those potential procedure-related benefits, “in the future there will also be opportunities in research and development, including data analytics and drug discovery and development,” predicted Philip W.Y. Chiu, MD, the director of the Endoscopy Center at Prince of Wales Hospital in Hong Kong. “On the physician side, AI will aid in patient monitoring after procedures, care coordination, the development of personalized treatment plans and administrative tasks. … Your endoscopy center as a whole will be data-driven by AI.”
—Caroline Helwick
Dr. Chiu reported financial relationships with Cornerstone Robotics and Henry Schein One. Dr. Cohen reported financial relationships with GI Windows, MD Medical Navigators, Micro-tech, Olympus, ROMTech and Virtual Health Partners. Dr. Hassan reported financial relationships with Boston Scientific, Fujifilm, Medtronic, Norgine, Odin and Olympus. Dr. Konda reported financial relationships with Ambu, Castle, Exact Sciences, Medtronic and Sebela. Dr. Ladabaum reported financial relationships with CheckCap, Freenome, Guardant, Kohler Ventures, Lean Medical, Medial EarlySign, Medtronic Neptune, Universal DX and Vivante. Dr. Wallace reported no relevant financial disclosures.
This article is from the September 2024 print issue.