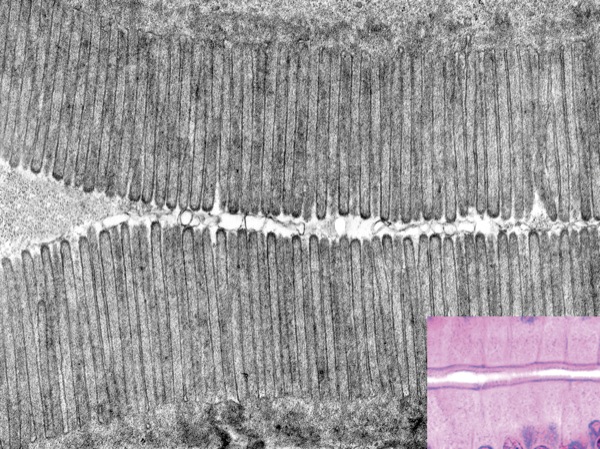
Barcelona, Spain—Microvilli may be small, but they tell many tales.
Two studies presented at the 2019 United European Gastroenterology Week found that the height of ileal epithelial microvilli can predict the likelihood that a patient with Crohn’s disease will respond to biologic therapy. Experts said the findings could lead to improvements in how clinicians manage patients with inflammatory bowel disease by optimizing drug treatment.
In one study (late-breaking abstract 3), Thaddeus Stappenbeck, MD, PhD, a professor of pathology and immunology at Washington University School of Medicine in St. Louis, and his colleagues retrospectively analyzed pre- and post-treatment histologic, endoscopic and clinical data from 95 people with Crohn’s disease who received either ustekinumab (Stelara, Janssen) or placebo as part of the UNITI-2 trial.
“What we found was that the greatest therapeutic effect and the greatest difference between treatment and placebo responders was seen in those with microvilli at least 1.7 microns in length,” Dr. Stappenbeck said.
Results showed that the rate of clinical response in the entire study population for ustekinumab and placebo was 65% and 39% at eight weeks, respectively. By subgroup, the rate among patients with microvilli height at least 1.7 microns was 85% and 20%, respectively (P=0.02). Conversely, among those with shorter microvilli, the gap in response was narrower, at 59% and 46% for ustekinumab and placebo recipients, respectively.
Patients with longer microvilli also had a numerically, but not significantly, higher likelihood of endoscopic response to ustekinumab at eight weeks (ustekinumab, 75%; placebo, 20%) than those with shorter microvilli (ustekinumab, 65%; placebo, 48%).
“Using an ileal biopsy and measuring microvilli length at baseline would be a helpful and easy-to-adopt predictor of clinical and endoscopic response,” Dr. Stappenbeck said.
In a separate study also presented at UEG Week (late-breaking abstract 4), combining ileal microvilli height with ileal epithelial cell death strongly identified responders to the biologic agent vedolizumab (Entyvio, Takeda).
The work followed on previous research by Julia Liu, MD, a staff physician at the University of Arkansas for Medical Sciences, in Little Rock, whose group found that death of ileal epithelial cells predicted response to vedolizumab, although specificity was low.
In the new study, Dr. Liu and her colleagues analyzed both cell death, defined as less than 14 activated caspase-positive cells per 1,000 intestinal epithelial cells, and microvilli height in 43 men and women with Crohn’s disease who were treated with vedolizumab at several centers.
Baseline demographic, medication and disease characteristics did not differ between the 58% of responders and the nonresponders. However, like Dr. Stappenbeck’s team, Dr. Liu’s group found that microvilli predicted response: 82% of patients with a mean height of ileal microvilli of 1.35 to 1.55 microns experienced a clinical response, compared with 44% of those with microvilli shorter than 1.35 microns and 40% of those with microvilli longer than 1.55 microns.
“We did expect that because vedolizumab and ustekinumab have different mechanisms of action, the optimal microvilli length range or cutpoints would be different,” Dr. Liu said, referring to the results of Dr. Stappenbeck’s study.
When Dr. Liu’s team focused on patients who met both the microvilli height and the definition of ileal epithelial cell death, they found that sensitivity was slightly lower but specificity rose: 78% of the patients meeting the two conditions experienced clinical response, compared with 25% of those not meeting either criterion.
“It would be interesting to examine whether altering microvilli length through diet or other approaches could help improve response to treatment,” Dr. Liu said, noting that a high-fat diet has been shown to reduce microvilli height in animals (J Nutr Sci Vitaminol 1994;40[2]:127-136).
Ashwin Ananthakrishnan, MD, MPH, an associate professor of medicine at Massachusetts General Hospital, in Boston, who was not involved in the studies, praised the investigators. “These are two very promising studies that contribute to an important area of research, namely being able to predict response to the expanding number of therapies with distinct mechanisms of action in IBD,” Dr. Ananthakrishnan told Gastroenterology & Endoscopy News.
Dr. Ananthakrishnan said future studies should try “to determine whether microvillus length is a predictor of response after adjusting for prior treatments and refractoriness to those treatments, which may themselves impact microvillus length.”
He added that further studies should analyze any correlations of microvilli height with other biomarkers, such as C-reactive protein and albumin, which sometimes are used to predict response to treatment.
—David Wild
Dr. Ananthakrishnan reported no relevant financial conflicts of interest. Dr. Liu reported holding a patent on an ileal pyroptosis stain assay. Dr. Stappenbeck reported research funding from Boehringer Ingelheim and Janssen.